Clinical applications of free medial tibial flap with posterior tibial artery for head and neck reconstruction after tumor resection
Introduction
Tumor resection causes damage in the head and neck which creates problems in swallowing, chewing, articulation, and vision, all of which seriously affect patients’ quality of life. The tissue types and content in the center face vary widely, and therefore the reconstruction of tissue defects should also vary. Presently, the main reconstruction approach is to use free tissue flaps including the fibula flap, the anterolateral thigh flap, and the rectus abdominis myocutaneous flap.
The radial forearm fasciocutaneous flap has been widely used for reconstruction since the 1980s (1) and it is a good choice for those who need a thin tissue flap to cover separated soft tissue defects. However, the donor sites of these flaps are located at exposed parts of the body and will leave behind detectable scars and pigmentation after surgery. A forearm flap from the flexor tendon zones is also a suitable choice for reconstruction. However, the activity of wrist is intense and frequent which can cause displacement between tendons to produce clefts in grafted skin and create complications like delayed wound healing, hand and wrist swelling, joint stiffness, and hand sensory abnormalities (2).
Due to the similarity in embryonic development, the upper and lower limbs have similar tissue composition, shape, and distribution of blood vessels and nerves. The medial limb area is usually covered with clothing which prevents exposure of scar and pigmentation and other aesthetic complications. Ankle joint mobility is relatively small, and there are fewer postoperative complications that can negatively affect the quality of life. In the past, the medial limb flap with the posterior tibial artery was commonly used for the repair of foot tissue defects, but its application for the head and neck reconstruction is relatively uncommon (3,4).
Cases and methods
Anatomy preparation
The posterior tibial artery is one of the branches of the popliteal artery. The upper part of the posterior tibial artery passes downwards to the gastrocnemius and soleus muscles. The lower part of posterior tibial artery runs down deep into the transverse fascia of the leg which splits from the intersection between the soleus muscle and the flexor digitorum. The posterior tibial artery splits at the medial malleolus into the anterior tibial artery and posterior tibial artery. The posterior tibial artery runs down with the tibial nerve and two veins. The upper, middle and lower parts of posterior tibial artery split muscular branches which perforate out a total of about 2 to 7 (most 4 to 5) cutaneous branches. The cutaneous branches are divided into the ascending branch and descending branch after piercing out from the deep fascia and connected beside the saphenous nerve to form longitudinal vascular chain. In the medial lower limbs, 3–4 cutaneous branches arise from the muscular branches of the posterior tibial artery that form the anatomical basis of the flap. The nutrient vessels of the skin at the medial lower limb mainly come from the saphenous artery, intermuscular branch of posterior tibial artery, and the anterior malleolus artery of the anterior tibial artery. The cutaneous branches of the posterior tibial artery form a plenty of anastomosis with the peripheral blood vessels and therefore the resection of flap does not affect the local blood supply.
Patient information
Nine patients were recruited from the Department of Otolaryngology Head and Neck Surgery, Beijing Tongren Hospital, Capital Medical University, between 2012 and 2016, including four males and five females with a median age of 50 (range, 14–58) years. Patients received surgical resection of the primary tumors. The primary tumor sites included nasal cavity and paranasal sinuses in two cases, oral cavity in four cases, hard palate in two cases, and lacrimal sac in one case. The pathological types were squamous cell carcinoma in six cases, mucoepidermoid carcinoma in one case, adenoid cystic carcinoma in one case, and clear cell carcinoma derived from salivary gland in one case. All patients received stage I repair after tumor resection (Table 1).
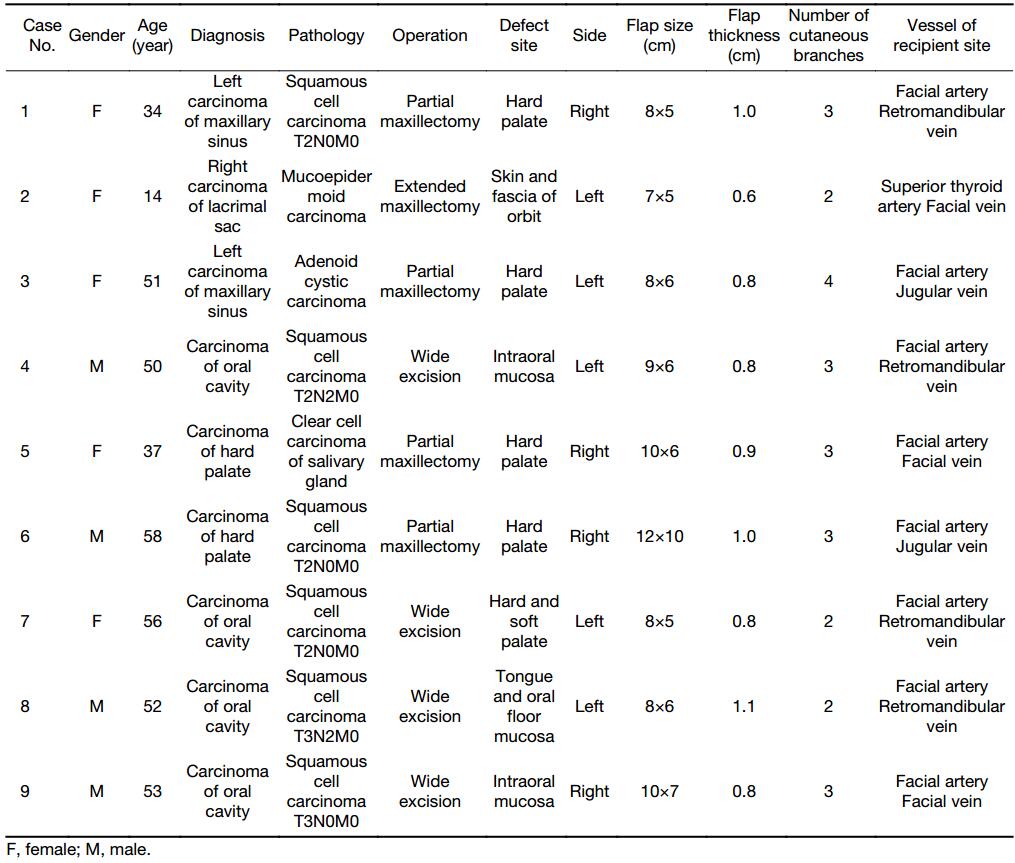
Full table
The study was approved by the Ethics Committee of Beijing Tongren Hospital, Capital Medical University, and written informed consent was obtained from all the participants.
Surgical methods
Preoperative preparation
In addition to the conventional preoperative preparation, we collected the complete medical history, including whether there was a trauma in the flap harvest site, as well as the surgical history, the presence of varicose veins, ischemic ulcers, an intermittent claudication, poor resistance to cold, and history of other peripheral vascular diseases. The physical examinations included the observation of venous drainage of the lower extremities to exclude the obstruction of superficial venous drainage, palpating the pulses of anterior tibial, posterior tibial, and dorsalis pedis artery, as well as the use of Doppler flow detector to check the shape, blood flow, and the number and location of the piercing branches. All patients did not perform preoperative angiography for the flap donor site.
Flap design
We used a Doppler flow detector to detect the location of the cutaneous branches of the posterior tibial artery. The flap was located at the axis connecting the medial condyle of tibia and medial malleolus. The size and location of flap were designed according to the extent of defect and the piercing out points of cutaneous branches. When a longer vascular pedicle is required, the lower piercing out point of posterior tibial artery branch can be selected. If a short vascular pedicle is needed, the flap should be as close as to the middle of the lower limb where the perforations are generally more abundant. The anterior and posterior edge of the flaps can reach the anterior and posterior median line, respectively.
Flap production and surgical operation
The patients took supine position with the knee flexion, mild external rotation and outreach of the hip, to fully expose the surgical site for the flap. A two-team operation was performed at head and neck and at flap harvest site, simultaneously. The anterior incision of designed flap was first made and the exact location and number of the cutaneous branches of posterior tibial artery were detected during the operation from the anterior malleolus to the fascia of long flexor digitorum. The posterior incision of the flap was then made. The cutaneous branches were detected under the deep fascia of the flexor digitorum and soleus. The posterior tibial artery, posterior tibial vein, and posterior tibial nerves beside the medial malleolus were found. After ligation of the distal posterior tibial artery and vein, the flap was flipped, and the deep fascia was sutured with edge of skin. We were careful to protect the cutaneous branches. The flap was resected under the deep fascia from down to up direction, and the vascular pedicle was separated upward along the interval between flexor digitorum longus muscle and soleus muscle. The long branches of posterior tibial artery that supply blood to the muscle and tibia were ligated and a long enough vascular pedicle was isolated. After ligating the proximal posterior tibial artery and vein, the flap was removed. The donor site was repaired using middle thick free abdominal flaps with local pressure bandage. Attention should be paid to pedis artery pulse. The posterior tibial arteries and veins were anastomosed with the corresponding vessels in the graft reception site. A 8-0 noninvasive nylon suture was used. Two patients used venous anastomosis (GEM Coupler, Synovis Micro Companies Alliance Inc., Birmingham, AL, USA). The drainage was placed after anastomosis. After the flap was folded and dressed according to the defect, the wound was sutured layer by layer.
Results
We used flaps with a length of 7–12 (average, 8.89) cm, width of 5–10 (average, 6.22) cm, and thickness of 0.6–1.0 (average, 0.87) cm. The number of the perforation vessels was 2–4 (average, 2.78). All the flaps survived completely with a satisfactory final shape. The wound at the head and neck healed smoothly in all patients and the sutures were removed 7–9 d after surgery. The donor site was implanted, and only a small amount of subcutaneous effusion occurred in two cases, and the wound healed after drainage and bandaging. The wounds at donor site in other patients healed well, and sutures were successfully removed 15–18 d after the surgery. We did not observe dysfunction in walking or ischemia in any patients. Also, no patients reported skin sensory abnormalities in their lower limbs. We also found that the load-bearing and walking capacity of the patients was not affected after a 3-month follow-up.
Typical cases
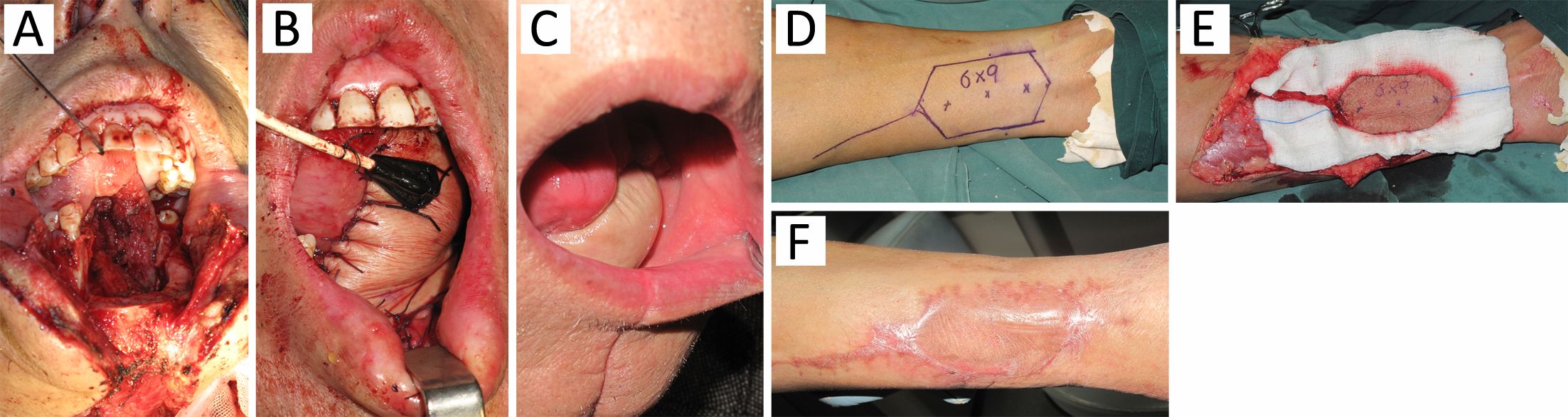
Case 4 was a 50 years old male who reported having a neck mass for one month. Physical examination found a painless mass with a size of about 3 cm × 2 cm × 2 cm at the left mandibular angle which was diagnosed as metastatic cancer through a biopsy. The examination also found a tumor at the mouth floor and a biopsy diagnosed it as a squamous cell carcinoma at T2N2M0. The patient accepted a mouth floor cancer resection plus bilateral neck lymph node dissection, and reconstruction with a left free medial tibial flap (9.0 cm × 6.0 cm × 0.8 cm) to cover the oral mucosal defect. The postoperative healing went well. Radiotherapy was performed (70 Gy) 6 weeks after the surgery (Figure 1). Patient had normal gait, and had no claudication or skin sensory abnormalities 3 months after surgery.
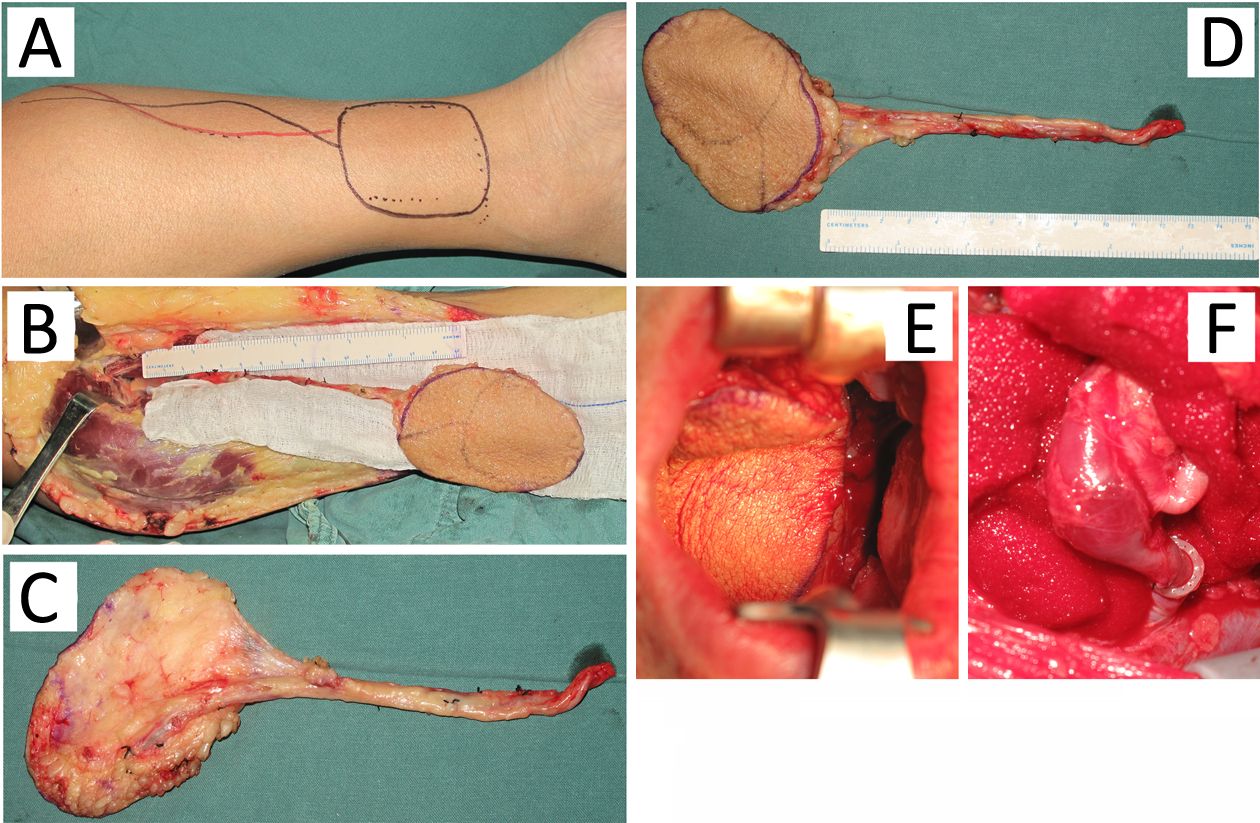
Case 6 was a 58 years old male who reported having palate pain and an ulcer for 2 months. A biopsy diagnosed as a moderately differentiated squamous cell carcinoma. No significant lymph node metastasis was found at the neck. The patient accepted a bilateral partial maxillary resection, and reconstruction with the right free medial tibial flap (12 cm × 10 cm × 1 cm). The nasal cavity was connected to the oral cavity so a folded flap was needed, which means that repair of the nasal cavity and oral cavity must happen simultaneously. Because of this, the final flap size was large (12 cm × 10 cm × 1 cm) with three cutaneous branches of the posterior tibial artery. Intraoperative venous anastomosis (GEM Coupler, Synovis Micro Companies Alliance Inc., Birmingham, AL, USA) was used and the postoperative healing was good (Figure 2). This patient took no postoperative radiotherapy.
Discussion
The medial tibial flap was first reported in the 1980s (5), and most studies used it as pedicled flap for proximal tissue defect repair in orthopedic problems (6,7). There are only few reports for its use in head and neck surgery (8). In contrast, the forearm flap has been widely applied to reconstruction of head and neck defects. The studies on anatomy, embryological development and clinical application suggest many similar features between tibial flap and the forearm flap, such as the high skin quality, thin subcutaneous fat, and softness. Like the forearm flap, the tibial flap can be designed according to different shapes and does not cause bloating after transplantation (9).
The tibial flaps have eight main advantages over the forearm flaps:
1) The flap donor site is hidden and does not affect the aesthetic appearance which is preferable to female patients. In this study, the ratio of male to female is 4:5.
2) The tibial flaps are tougher and more durable than the forearm flaps, which makes them more suitable for repairing the defects in tongue, mouth floor, buccal and hard palate.
3) the limb is longer than the forearm, and the vascular pedicle could be longer. William et al. reported that the average length of tibial artery pedicle can be up to 17.60±1.93 cm (9).
4) The diameter of posterior tibial artery is equivalent to the diameter of facial artery, while the posterior tibial artery is anatomically constant with less variation. Thus, posterior tibial arteries are easier to match and the success rate is high. In this study, the facial artery was successful anastomosed with the cutaneous branches of the posterior tibial artery in six of the seven patients.
5) The lower limb is thicker than forearm, and has more abundant soft tissue, while the posterior tibial artery supplies an area up to 194 cm2. In this study, the largest flap in one patient was up to 12 cm × 10 cm.
6) The posterior tibial artery is only one of the three arteries to supply blood in lower limb and not the major feeding vessel. Thus, resecting the flap may not affect the lower limb’s functions. In our study, no ischemia, claudication, skin sensory abnormalities, lower extremity load, or joint movement and walking were found in the lower limb.
7) The musculus medialis of lower limb is more developed than that of the forearm, and the skin graft can be implanted on muscle without affecting the tendon. In addition, the ankle has a good stability and relatively small activity, and the displacement between tendons is small in lower limb than that in the forearm. This type of graft is less prone to postoperative skin rupture, and wound healing delay.
8) The lower limbs are far away from the head and neck, and two-group operations can be performed simultaneously in one patient.
Although we do not observe serious donor-site morbidity, we recognize that the posterior tibial artery is one of the three arteries of the lower limb and the foot which can be detrimental if damaged. The sacrifice of posterior tibial artery is not harmful in theory because the human body can quickly establish a collateral circulation through the rich network of blood vessels in lower limb to provide adequate blood supply. However, the distal limb ischemia is a main concern for clinicians and researchers. Thus, the surgical indications and the choice of patients are the key to a successful operation. In this study, the medical history was collected, and physical examination and ultrasound examination were performed before surgery to exclude the lower limb vascular disease to protect the blood supply of the donor site.
Conclusions
The free medial tibial flap with cutaneous branches of the posterior tibial artery is easy to make, with a relatively constant anatomical position, and a canonical diameter and pedicle length that are suitable for the repair of head and neck tumor defects. The subcutaneous fat is thin, the skin flap is soft, and the donor site is far away from the reconstruction site. The donor site is hidden and causes less damage, and has a better propensity to repair defects requiring thin-layer epithelium to cover or separated soft tissue defects. Thus, the medial tibial flap is superior to the forearm flap in many respects and can be used for the repair in head and neck surgery.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Yang GF, Chen PJ, Gao YZ, et al. Forearm free skin flap transplantation: a report of 56 cases.1981. Br J Plast Surg 1997;50:162–5. [PubMed]
- Ng RW, Chan JY, Mok V, et al. Free posterior tibial flap for head and neck reconstruction after tumor expiration. Laryngoscope 2008;118:216–21. [PubMed] DOI:10.1097/MLG.0b013e3181593dcb
- Steel BJ, Cope MR. A brief history of vascularized free flaps in the oral and maxillofacial region. J Oral Maxillofac Surg 2015;73:786.e1–11. [PubMed] DOI:10.1016/j.joms.2014.12.005
- Xu ZF, Shang DH, Duan WY, et al. Free posterior tibial artery perforator flap for floor of mouth reconstruction: a case report. Microsurgery 2011;31:659–61. [PubMed] DOI:10.1002/micr.20931
- Hwang WY, Chen SZ, Han LY, et al. Medial leg skin flap: vascular anatomy and clinical applications. Ann Plast Surg 1985;15:489–91. [PubMed]
- Wu H, Sheng JG, Zhang CQ. Free medial plantar flap connection with a posterior tibial artery flap in reconstruction of fore-mid foot skin defect. Plast Reconstr Surg Glob Open 2016;4:e1091. [PubMed] DOI:10.1097/GOX.0000000000001091
- Jing-Chun Z, Kai S, Jia-Ao Y, et al. Large heel soft tissue defects managed successfully with reverse medial crural fasciocutaneous flap: a 7-year single-center experience with 21 consecutive cases. J Plast Reconstr Aesthet Surg 2015;68:40–8. [PubMed] DOI:10.1016/j.bjps.2014.09.041
- Chen HC, Tang YB, Noordhoff MS. Posterior tibial artery flap for reconstruction of the esophagus. Plast Reconstr Surg 1991;88:980–6. [PubMed]
- Chan YW, Ng RW, Wei WI. Anatomical study and clinical applications of free posterior tibial flap in the head and neck region. Plast Reconstr Surg 2011;128:131e–139e. [PubMed] DOI:10.1097/PRS.0b013e318221db67
