Comparison of survival outcomes between transthoracic and transabdominal surgical approaches in patients with Siewert-II/III esophagogastric junction adenocarcinoma: a single-institution retrospective cohort study
Weihan Zhang1,2, Xinzu Chen1,2, Kai Liu1,2, Kun Yang1,2, Xiaolong Chen1,2, Ying Zhao1,3, Yongfan Zhao4, Jiaping Chen1, Longqi Chen4, Jiankun Hu1,2
Abstract
Objective: To compare the survival outcomes of transabdominal (TA) and transthoracic (TT) surgical approaches in patients with Siewert-II/III esophagogastric junction adenocarcinoma.
Methods: This retrospective study was conducted in patients with Siewert-II/III esophagogastric junction adenocarcinoma who underwent either TT or TA operations in the West China Hospital between January 2006 and December 2009.
Results: A total of 308 patients (109 in the TT and 199 in the TA groups) were included in this study with a follow-up rate of 87.3%. The median (P25, P75) number of harvested perigastric lymph nodes was 8 (5, 10) in the TT group and 23 (16, 34) in the TA group (P<0.001), and the number of positive perigastric lymph nodes was 2 (0, 5) in the TT group and 3 (1, 8) in the TA group (P<0.004). The 5-year overall survival (OS) rate was 36% in the TT group and 51% in the TA group (P=0.005). Subgroup analysis by Siewert classification showed that 5-year OS rates for patients with Siewert II tumors were 38% and 48% in TT and TA groups, respectively (P=0.134), whereas the 5-year OS rate for patients with Siewert III tumors was significantly lower in the TT group than that in the TA group (33% vs. 53%; P=0.010). Multivariate analysis indicated that N2 and N3 stages, R1/R2 resection and a TT surgical approach were prognostic factors for poor OS.
Conclusions: Improved perigastric lymph node dissection may be the main reason for better survival outcomes observed with a TA gastrectomy approach than with TT gastrectomy for Siewert III tumor patients.
Keywords: Siewert classification; adenocarcinoma of esophagogastric junction; transthoracic; transabdominal; prognosis
Submitted Jan 31, 2016. Accepted for publication Mar 21, 2016.
doi: 10.21147/j.issn.1000-9604.2016.04.04
Introduction
Although the incidence of gastric cancer is declining, the incidence of esophagogastric junction (EGJ) tumors is increasing (1-3), and these trends are evident both in the East Asian and the Western countries (4-6). According to the Siewert classification system, Siewert type I tumors are defined as adenocarcinomas of the distal esophagus with a center located within 1-5 cm above the anatomic EGJ; Siewert type II tumors are true carcinomas of the cardia with a tumor center within 1 cm above and 2 cm below the EGJ; and Siewert type III tumors include subcardial carcinomas with centers between 2-5 cm below the EGJ (5). Thus far, much controversy has centered on the proper surgical approaches of these tumors. Gradually, previous studies concluded that EGJ tumors should be treated independently from gastric and esophageal cancers (7, 8). For Siewert I tumors, due to the mediastinal lymph node metastasis (9), transthoracic (TT) surgery can achieve better survival outcomes than non-TT approaches (10, 11). However, because Siewert type II/III tumors had a lower chance of mediastinal lymph node metastasis than Siewert type I tumors, thoracic incision surgery is not preferred in these cases (12-14). To investigate the survival outcomes among different surgical approaches in patients with Siewert type II/III tumors, we retrospectively analyzed those patients who were diagnosed with Siewert type II/III tumors and underwent abdominal or thoracic surgery in the Department of Gastrointestinal Surgery or the Department of Thoracic Surgery in the West China Hospital, Sichuan University, China.
Materials and methods
Patients
With the approval of Biomedical Ethics Committee of West China Hospital, Sichuan University, data from Siewert type II/III tumor patients who underwent transabdominal (TA) and TT surgery from January 2006 to December 2009 were retrospectively collected from the database of West China Hospital, Sichuan University. The Department of Thoracic Surgery was responsible for thoracic incision surgery and the Department of Gastrointestinal Surgery was responsible for abdominal incision surgery. The inclusion criteria were: 1) Siewert II/III adenocarcinoma; 2) purely TT or TA approaches with total or proximal gastrectomy; 3) no distal metastasis; 4) invasive esophageal tumor length less than 3 cm; and 5) complete medical records available. The exclusion criteria were: 1) remnant stomach cancer and non-epithelial malignant tumors; or 2) other malignant diseases. A total of 308 patients (109 patients in the TT group and 199 patients in the TA group) were included in the study and grouped according to their respective surgical approaches. No patients underwent neoadjuvant chemotherapy during the study period. In addition, patients in the TT group underwent purely TT surgery, and neither the TT nor TA group included patients treated with thoracoabdominal surgical approaches.
Siewert patient classifications
Measuring the distance from the tumor center to the anatomic cardia determined the Siewert subtypes. Gastrointestinal radiography and upper gastrointestinal endoscopy were used preoperatively to evaluate the Siewert subtypes. If a Siewert type I subtype tumor was suspected during the preoperative evaluation, either TT or thoracoabdominal incision surgery was performed. Those patients who were preoperatively evaluated as having Siewert II/III tumors underwent TA or TT surgery, and the Siewert subtype was confirmed upon intraoperative evaluation.
Treatment
Patients in the TT group received gastrectomy exclusively by a left thoracic approach, and patients in the TA group received gastrectomy through abdominal incision. Only thoracic surgeons of the Thoracic Surgery Department performed TT surgeries, and only gastrointestinal surgeons in the Gastrointestinal Surgery Department performed TA surgeries. For the resection patterns, tumor characteristics as well as the surgeon's preference and habits dictated whether a total or proximal gastrectomy was necessary for patients in the TA group. However, because there was no thoracoabdominal incision surgery in this study, no patients in the TT group underwent total gastrectomy. Lymph node dissections along the left gastric artery, celiac artery and common hepatic artery were completed during either of the above surgical approaches. Lymphadenectomies in the TA group were classified according to the criteria of the Japanese Gastric Cancer Association (JGCA) (15). Lymph nodes along with the splenic artery were dissected only for patients in the TA group, and the mediastinal lymph nodes dissected only for patients in the TT group. Specifically, lymphadenectomy in the TT group included lymph nodes of the diaphragm, lower esophagus and peritracheal tissues. For the reconstruction of the digestive tract, total gastrectomy in the TA group was accompanied by esophagojejunal Roux-en-Y anastomosis. In all partial gastrectomy, esophageal-gastric anastomosis was used. Anastomosis was performed in the thoracic cavity in the TT group and under the esophageal hiatus in the TA group. All anastomoses from the two groups were completed with the circular mechanical stapler and reinforced by hand-sewn sutures.
The postoperative adjuvant chemotherapy was recommended for those patients with disease advanced beyond the T2 stage or with lymph node metastasis in any T stage. Combinations of fluoropyrimidine and platinum regimens were used as the first-line postoperative chemotherapy treatment strategies.
Histopathological assessment
Experienced pathologists in the West China Hospital, Sichuan University assessed all histopathological slides. Removed lymph nodes were counted and assessed separately. Pathologic information included histologic differentiation (grade), depth of tumor invasion (T stage), regional lymph-node status (N stage), the number of lymph nodes (positive and harvested), lymph node macroscopic type and the degree of resection (R0, no residual tumor; R1, microscopic residual tumor; R2, macroscopic residual tumor).
Long-term follow-up
Regular postoperative follow-up strategy (at least twice per year during the first two years and at least once per year till the last year) was recommended to all of the patients underwent either thoracic or abdominal surgery. The details of the follow-up strategies included: 1) physical examination, routine blood examination, liver function test and serum tumor biomarkers test every three or six months; 2) thoracic and abdominal enhanced computed tomography and upper gastrointestinal endoscopy at least once per year; and 3) once metastasis was suspected, further examination would be performed. The follow-up information was updated by January 1, 2015. In the 308 patients, 39 patients lost contact during follow-up, and the total follow-up rate was 87.3% with a median duration of 44 (2-108) months. Contact information changes (telephone number and address) and refusal to participate in hospital interviews were the main reasons for follow-up loss.
Statistical analysis
IBM SPSS Statistics (Version 19.0, IBM Corp., New York, USA) was used for statistical analysis. Continuous data with non-normal distribution were described by the median (P25, P75). For categorical data, rates or proportions were used. Continuous data with non-normal distributions were analyzed by the Mann-Whitney U test. For comparisons among categorical data, the Chi-square test or the Fisher's exact test was used for the unordered categorical data and the Mann-Whitney U test was used for the ordinal categorical data. Survival analyses were performed in patients with complete follow-up information. Kaplan-Meier curves (log-rank test) were used for the analyses of survival outcomes, and the log-rank test was performed to assess statistical significance. The 5-year overall survival (OS) rate was calculated by the life-table test. Multivariate adjusted factor analysis was performed using the Cox proportional hazard modeling. Two-tailed P values of less than 0.05 were considered statistically significant.
Results
Patient characteristics
Three hundred and eight patients were enrolled in this study, including 109 patients in the TT group, and 199 patients in the TA group. The clinicopathological characteristics were compared between the two groups (Table 1). Gender proportion and mean age were similar between the two groups. The proportions of Siewert type II and type III tumors between the TT and the TA groups were 69/40 and 101/98 (P=0.034), respectively. There were no significant differences between the two groups with regards to body mass index (BMI), tumor size, histological grade, T stage, N stage or TNM stage (P>0.05).
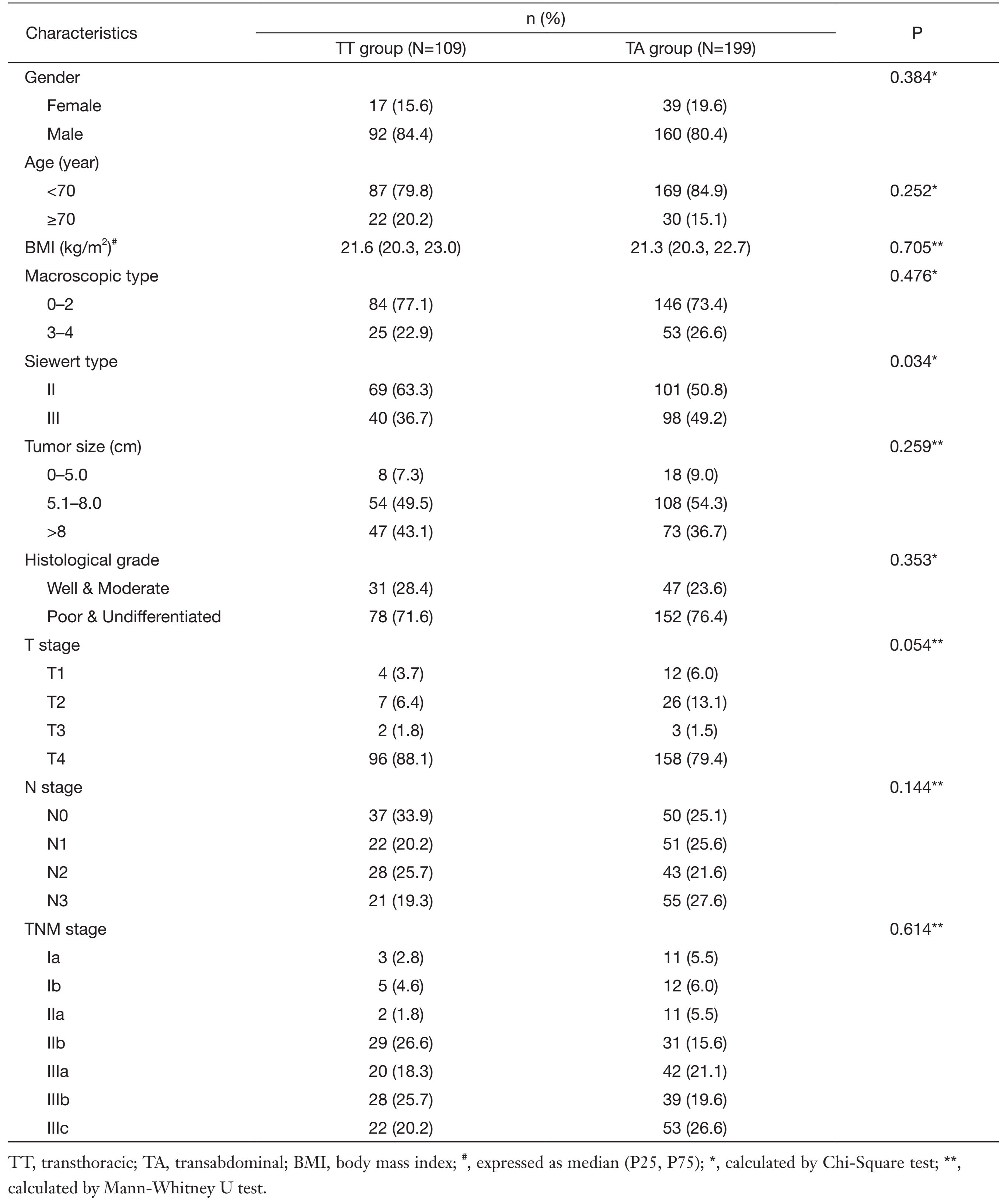
Full table
Operative variables
The operative variables are listed in Table 2. The average surgery duration of the TA group was significantly longer than that of the TT group [215.0 (172.5, 252.5) vs. 240.0 (205.0, 285.0), respectively, P<0.001]. The median number of total harvested lymph nodes was 10 (6, 15) in the TT group and 23 (16, 35) in the TA group (P<0.001), and of these, the total number of positive lymph nodes was 2 (0, 15) in the TT group and 2 (0, 8)in the TA group (P=0.025). In the subgroup analysis of the perigastric lymph nodes, the number of harvested perigastric lymph nodes was 8 (5, 10) in the TT group and 23 (16, 34) in the TA group (P<0.001), and the number of positive perigastric lymph nodes was 8 (5, 10) in the TT group and 23 (16, 34) in the TA group (P=0.004). There was no difference in residual tumor degree according to the JGCA gastric cancer classification between the two groups, and the curative resection rate (R0) was 94.5% in the TT group and 97.0% in the TA group (P=0.357).
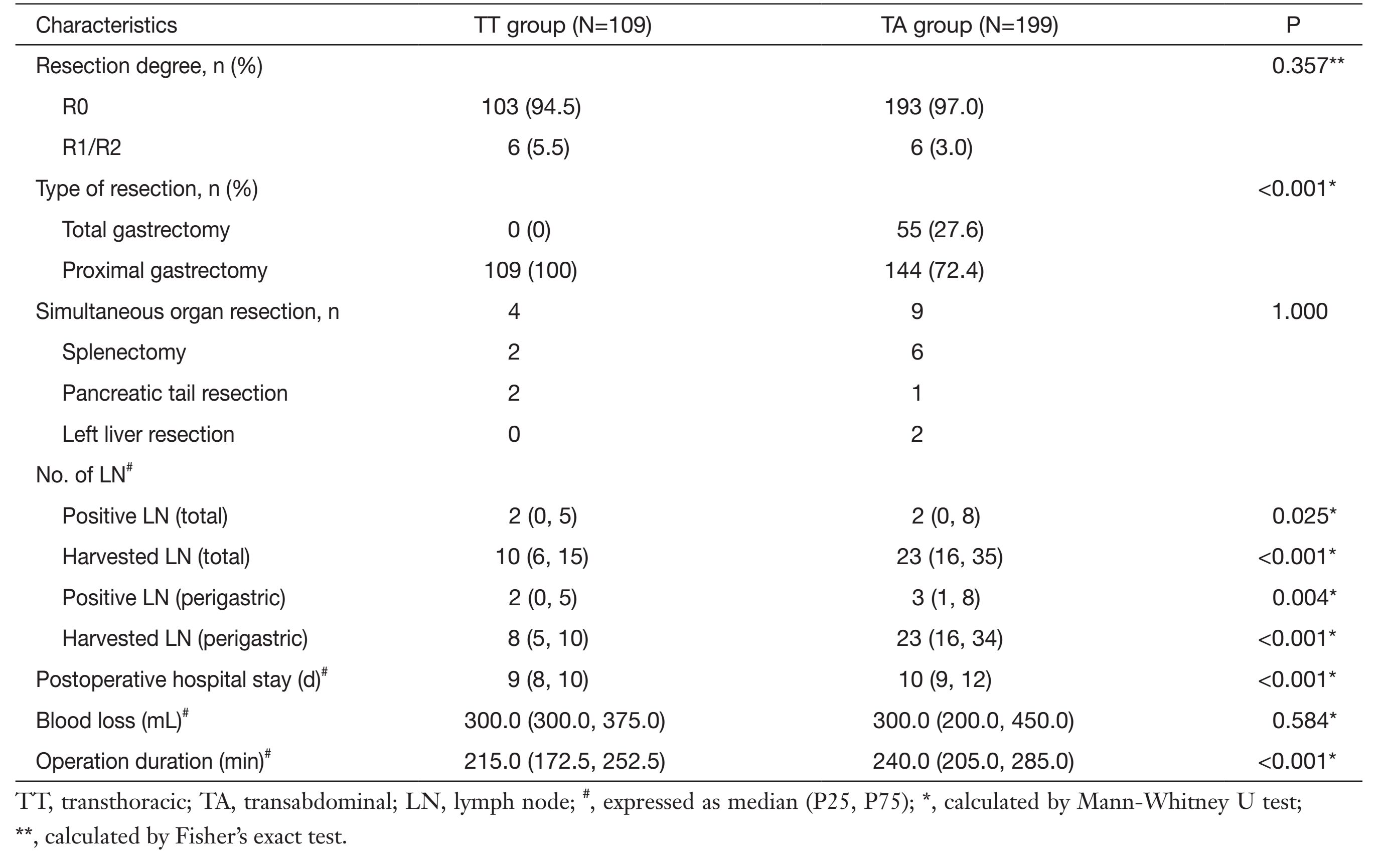
Full table
Morbidity and mortality
There were no intraoperative mortalities in either of the two groups. The postoperative hospital stay was shorter for patients in the TT group than those in the TA group [9 (8, 10) d vs. 10 (9, 12) d, P<0.001]. Postoperative morbidity and mortality of the two groups 30 d post-surgery were comparable (Table 3). Two patients in the TA group died during the first 30 d after surgery: one was due to the postoperative acute respiratory failure and the other was severe intraperitoneal bleeding (P=0.541).
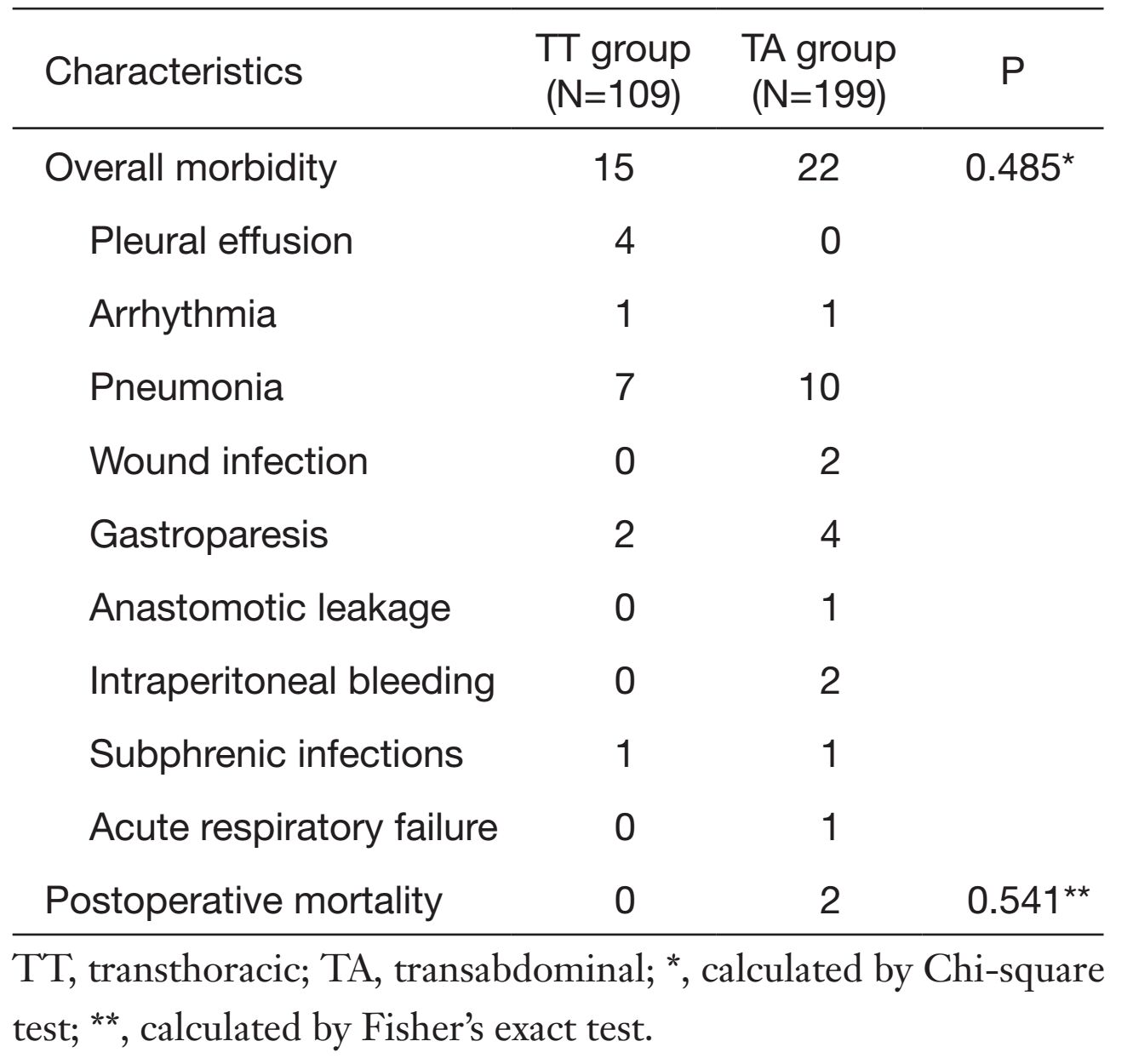
Full table
Long-term survival and prognostic factors
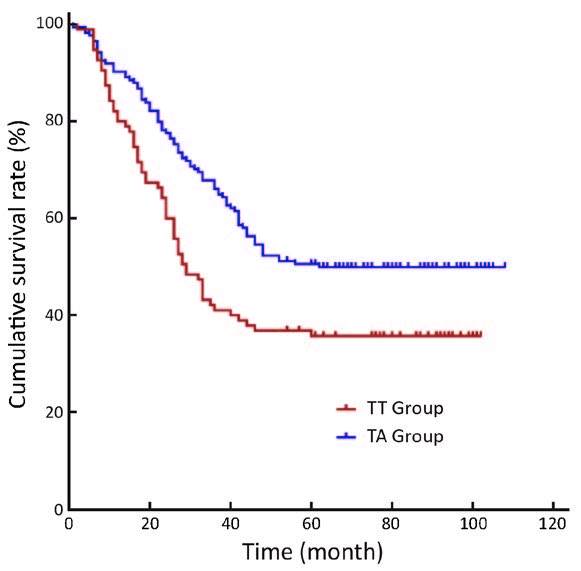
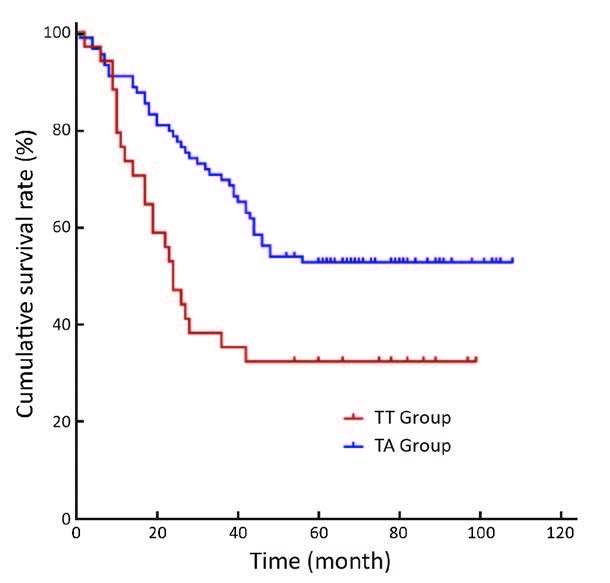
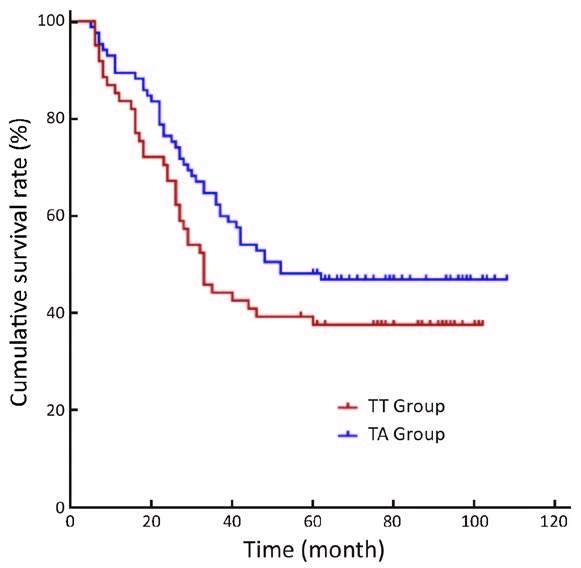
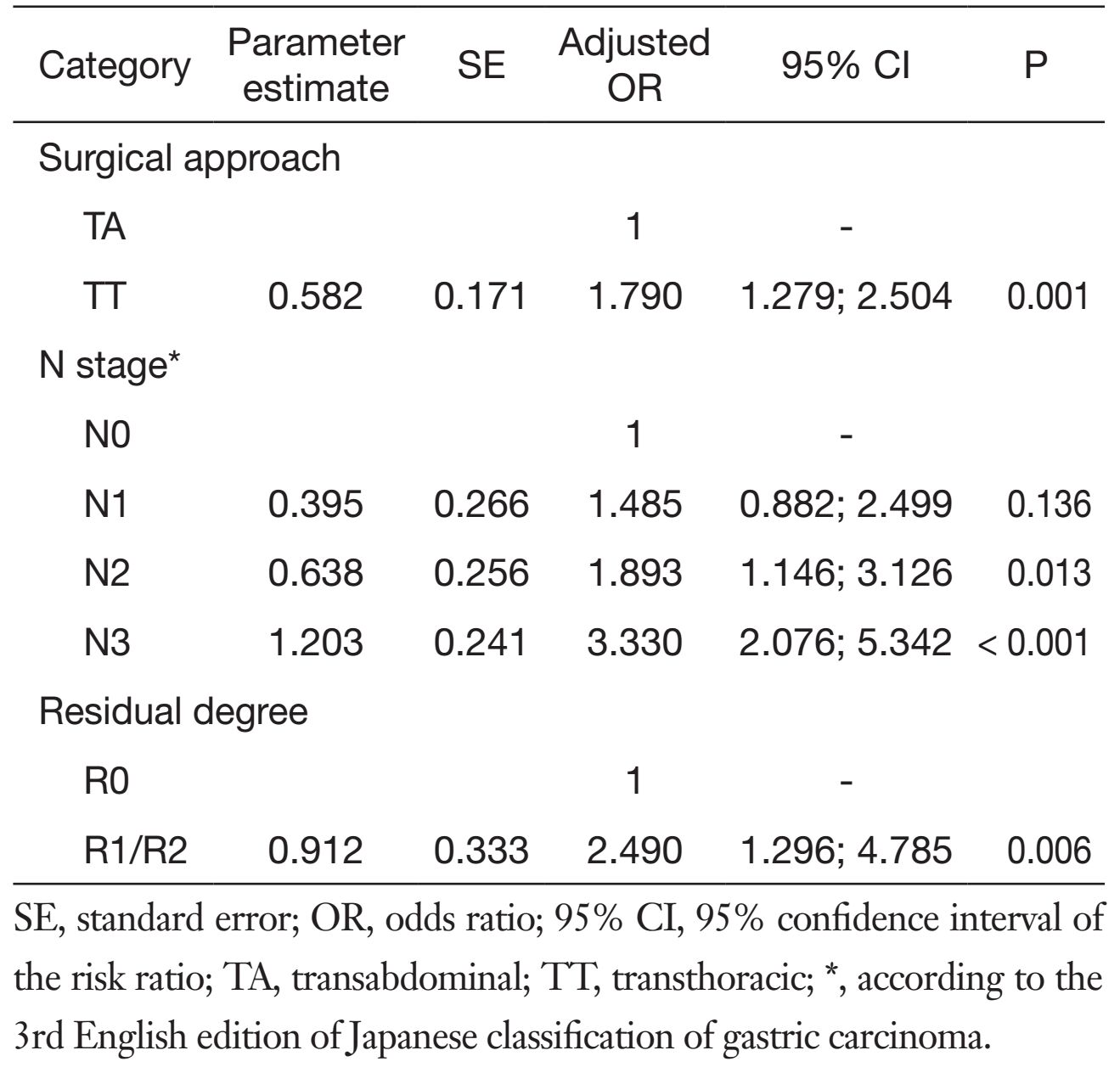
For the long-term survival outcomes, the survival curves of the two groups are presented in Figures 1, 2, 3. The 5-year OS rates were 36% in the TT group and 51% in the TA group (log-rank test, P=0.005). Subgroup analysis showed that Siewert type III tumor patients in the TA group had significantly better 5-year OS rates than patients in the TT group (33% vs. 53%, log-rank test, P=0.010). For the Siewert type II tumor patients, the 5-year OS rate was 38% in the TT group and 48% in the TA group (log-rank test, P=0.134). Univariate analysis revealed that the factors impacting the prognosis were surgical approach (P=0.005), tumor size (P=0.029), histological grade (P=0.045), residual degree (P<0.001), T stage (P=0.008), and N stage (P<0.001). Cox multivariate analysis demonstrated that the surgical approach [TA vs. TT, hazard ratio (HR), 1:1.790; 95% confidence interval (95% CI), 1.279-2.504; P=0.001], N1 stage [N0 vs. N1, HR, 1:1.485; 95% CI, 0.882-2.499; P=0.136], N2 stage (N0 vs. N2, HR, 1:1.893; 95% CI, 1.146-3.126; P=0.013), N3 stage (N0 vs. N3, HR, 1:3.330; 95% CI, 2.076-5.342; P<0.001) and residual degree (R0 vs. R1/R2, HR, 1:2.490; 95% CI, 1.296-4.785; P=0.006) were independently associated with OS in patients with Siewert II/III adenocarcinomas (Table 4).
Full table
Discussion
EGJ tumor patients have poor survival outcomes, especially those with Siewert type II and III tumors (9, 16). Although efforts had been dedicated to improving treatment of these tumors, controversies regarding the best surgical approaches for these patients persist. Thoracic surgeons deem these tumors to be similar to those observed in cases of esophageal cancer, and thus, thoracic surgery is the best approach. On the other hand, gastrointestinal surgeons regard abdominal approach surgery to be the primary option for Siewert II/III tumors. Therefore, we conducted this retrospective study to determine which TA surgical approach was the better option for the Siewert II/III tumor patients compared with TT surgical approaches, and found out that TA gastrectomy had more complete dissection of abdominal lymph nodes and better OS outcomes than TT surgery.
Siewert recommended that tailored surgical treatment strategies be necessary to treat this disease (9). Meanwhile, some researchers have observed that Siewert type II/III tumors should be treated by abdominal incision gastrectomy rather than thoracic approaches due to improved survival outcomes (17). A previous study found that the postoperative mortality and morbidity rates were significantly higher when patients were treated with TT approaches than that when patients were treated with the abdominal incision surgery (11, 17). However, some studies observed that TT approach operation did not result in increased mortality and morbidity compared with TA approach surgery (9).
According to the Japanese gastric cancer treatment guidelines for resection patterns, proximal gastrectomy is suitable for those lesions invading only the mucosa and submucosa (T1), whereas total gastrectomy is the standard surgical treatment option for advanced gastric carcinomas (T2-T4). For lesions located in the upper third of the stomach, total gastrectomy had lower recurrence rates than proximal gastrectomy (18, 19). In this study, patients in the TT group underwent only proximal gastrectomy, whereas patients in the TA group underwent both proximal gastrectomy and total gastrectomy. In this study, we found that the 5-year OS rate was 46% for patients who received proximal gastrectomy and 41% for patients with total gastrectomy (P=0.408). Patients with large tumors underwent total gastrectomy as performed by gastrointestinal surgeons, and the tumor characteristics and other surgical factors may explain the similar survival outcomes between the two resection patterns observed in this study. However, the total gastrectomy with D2 lymphadenectomy is the standard treatment strategy for the advanced upper third gastric cancers according to the Japanese classification (20). Therefore, it must be emphasized here that the high proportion of proximal gastrectomy in this study is inappropriate according to today's guidelines.
Lymph node metastasis is one of the most important prognostic factors for gastric cancer that may significantly influence survival outcomes. D2 lymphadenectomy, which is recommended by the JGCA in 2010, is the internationally recognized standard (20). For the low mediastinal lymph nodes (No.110 and No.111), some researchers concluded that dissection was necessary in all 3 Siewert subtype tumors. However, Yamamoto et al. observed that the rates of mediastinal lymph node metastasis were only 7.5% for patients with Siewert type II tumors and 2.8% for those with Siewert type III tumors (21). In addition, a previous Korean study observed that Siewert type II/III tumors might be successfully treated by total abdominal gastrectomy without mediastinal lymph node dissection (22). For the perigastric lymph nodes, a previous study found that the rates of perigastric lymph node metastasis were 32.9% in Siewert type II tumors and 50% in Siewert type III tumors (23). In this study, we did not compare metastasis rates for each lymph node location due to different lymphadenectomy strategies pursued in the two groups, but we observed that the total number of collected lymph nodes was 10 (6, 15) and 23 (16, 35) (P<0.001) and the number of positive lymph nodes was 2 (0, 5) and 2 (0, 8) (P=0.008) in the TT group and in the TA group, respectively. Furthermore, when perigastric lymph node dissections were compared between the two groups, the TA group had increased number of harvested and positive lymph nodes compared with the TT group in this study (P<0.001). For the survival analysis, we found that advanced N stage disease (N2 and N3 stages) and surgical approaches were risk factors for the poor prognosis. However, the two surgical approaches have totally different lymphadenectomy strategies. Thoracic surgeons usually place a greater emphasis on the lymphadenectomy in the thoracic cavity, whereas gastrointestinal surgeons pay more attention to the perigastric lymphadenectomy for adenocarcinomas of the EGJ. However, TA surgery can result in an increased number of harvested perigastric lymph nodes, which will also increase the number of positive lymph nodes compared with those obtained from the TT surgery. On the other hand, the No.7, No.8, No.9 nodes along the celiac trunk and its branches are frequently involved in the Siewert II/III tumors (24), complete resection of these nodes by pure TT surgery is difficult and the remaining of potential positive lymph nodes may lead to tumor recurrence and cancer-related death. Therefore, complete perigastric lymphadenectomy of abdominal approach may be a major reason for the improved survival outcomes observed in this study.
Another important factor considered by surgeons is the resection degree as all surgical oncologists strive to leave no residual tumor cells after surgery. Positive resection margin is an independent risk factor for gastric cancer patient survival (25). Abdominal incision surgery cannot achieve a similar resection degree to thoracic surgery for Siewert type I tumors (26), and for the Siewert type II/III tumors, abdominal incision surgery can result in similar resection degrees to those obtained with left thoracic-abdominal approaches (9). Our study observed that thoracic and abdominal incision surgeries can achieve similar resection degrees and that the resection degree is another important prognostic factor.
Moreover, we observed that there were more Siewert type III tumor patients in the TA group than that in the TT group. Were the differences in survival outcomes due to the different Siewert proportions between the two groups? Although a previous study indicated that Siewert II tumors had completely different tumor characteristics from those of Siewert III tumors (27), a separate study reported that Siewert II and Siewert III tumors have similar characteristics (22). In addition, according to the results of our previous study, perigastric lymph node metastasis was the major metastasis outcome for both the Siewert II and Siewert III tumors, and these two tumors had similar prognoses by abdominal surgery (24). On the one hand, in this study, subgroup analysis found that Siewert III patients in the TA group had significant OS benefits compared with those in the TT group (P=0.010). No significant differences between Siewert II tumor patients in these two groups were observed (P=0.134). Therefore, abdominal surgery may be the primary therapeutic option for patients with Siewert III tumors. On the other hand, we are also aware that the lack of transhiatal surgeries in the TA group is one of the limitations in this study. Consequently, whether purely thoracic, thoracoabdominal or abdominal incision surgery (include the transhiatal surgery) is the best surgical therapeutic option for Siewert II tumors requires further study.
Last, another factor that may influence the choice of the resection patterns and the surgical approach pursued may be the BMI. Although some studies found that obesity is not a risk factor for postoperative complications (28), obesity may pose significant operative challenges for the surgeon and increase the risk of anastomotic leakage during both transhiatal and thoracic surgical approaches (29, 30). Therefore, more extensive esophageal resection via abdominal surgery may be impossible for patients with high BMIs. For tumors invading less than 3 cm of the distal esophagus, the Japanese guidelines recommend transhiatal surgical approach. However, the BMI levels are substantially different between patients from Eastern to Western countries, and whether the recommendation of 3 cm is safe and efficacious for Western patients requires further study. It is our opinion that the specific tumor length invading the distal esophagus and patients' individual characteristic should determine whether abdominal incision surgery is pursued in patients from Western countries.
Our study has some limitations. First, this study lacked data regarding lymph node metastasis status at each location, but the number of harvested and positive lymph nodes (total and perigastric) revealed that TA approaches could achieve more complete perigastric lymphadenectomy. Second, this was a retrospective study with relatively small sample size, and the rate of loss to follow-up (12.6%) in this study is greater than 10%, which may influence the final survival results. Third, because of the natural limitations of a retrospective study, this study contained a selection bias, and did not analyze the relationship between preoperative variables and surgical approaches. In this study, surgical approaches are mostly decided by the preference and selection of surgeons and patients. Generally, thoracic surgeons preferred and performed thoracic incision surgery whereas gastrointestinal surgeons preferred and performed abdominal surgery. Therefore, the selection of surgical approach is the major bias in this study. In order to minimize the bias, we only collected consecutive patients with length of tumor infiltration in esophagus less than 3 cm to compare the two approaches. Fourth, in this study, the proportion of proximal gastrectomy is relatively high in consideration of the tumor stages, which is an inappropriate and non-standard treatment from today's vantage point. According to the recent Japanese guidelines, it needs to be emphasized that total gastrectomy is the best choice for the advanced Siewert II/III tumors.
However, we investigated these non-standard treatments in recent years and conducted this study with the aim to report the previous situations regarding the treatment of EGJ tumors at a high-volume Chinese medical center. Currently, we have made some changes according to the Japanese guidelines and the results of JCOG9502 (13, 17). Siewert II/III patients with esophagus invasion length less than 3 cm underwent TA (included transhiatal) approach surgery performed by the gastric cancer multidisciplinary team of West China Hospital. Despite these limitations, this study successfully demonstrated that TA surgery can ensure more complete dissection of abdominal lymph nodes and a better OS outcome than TT surgery.
Conclusions
Briefly, for Siewert type II/III tumors, TA surgery resulted in complete perigastric lymphadenectomy and increased the number of positive and negative lymph nodes. TA surgery can attain better prognosis than the purely TT surgery for Siewert III tumors. Therefore, TA gastrectomy may be a better therapeutic option than TT gastrectomy for patients with Siewert type III tumors.
Acknowledgements
The authors thank the substantial work of Volunteer Team of Gastric Cancer Surgery (VOLTGA) based on Multidisciplinary Team (MDT) of Gastrointestinal Tumors, West China Hospital, Sichuan University, China. The authors also sincerely appreciate Dr. Zhong Q. Wang from Pennington Biomedical Research Center, Louisiana State University System, USA for his English language support.
Funding: This work was supported by National Natural Science Foundation of China (No. 81372344).
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Colquhoun A, Arnold M, Ferlay J, et al. Global patterns of cardia and non-cardia gastric cancer incidence in 2012. Gut 2015;64:1881–8. [PubMed] DOI:10.1136/gutjnl-2014-308915
- Chen W, Zheng R, Baade PD, et al. Cancer statistics in China, 2015. CA Cancer J Clin 2016;66:115–32. [PubMed] DOI:10.3322/caac.21338
- Chen W, Zheng R, Zeng H, et al. Annual report on status of cancer in China, 2011. Chin J Cancer Res 2015;27:2–12. [PubMed]
- Deans C, Yeo MS, Soe MY, et al. Cancer of the gastric cardia is rising in incidence in an Asian population and is associated with adverse outcome. World J Surg 2011;35:617–24. [PubMed] DOI:10.1007/s00268-010-0935-0
- Siewert JR, Stein HJ. Classification of adenocarcinoma of the oesophagogastric junction. Br J Surg 1998;85:1457–9. [PubMed] DOI:10.1046/j.1365-2168.1998.00940.x
- Liu K, Yang K, Zhang W, et al. Changes of esophago-gastric junctional adenocarcinoma and gastroesophageal reflux disease among surgical patients during 1988-2012: A single-institution, high-volume experience in China. Ann Surg 2016;263:88–95. [PubMed] DOI:10.1097/SLA.cjcr-28-4-41301148
- Chen XZ, Zhang WH, Hu JK. Lymph node metastasis and lymphadenectomy of resectable adenocarcinoma of the esophagogastric junction. Chin J Cancer Res 2014;26:237–42. [PubMed]
- Kurokawa Y, Sasako M, Doki Y. Treatment approaches to esophagogastric junction tumors. Dig Surg 2013;30:169–73. [PubMed] DOI:10.1159/000350880
- Rüdiger Siewert J, Feith M, Werner M, et al. Adeno-carcinoma of the esophagogastric junction: results of surgical therapy based on anatomical/topographic classification in 1, 002 consecutive patients. Ann Surg 2000;232:353–61. [PubMed] DOI:10.1097/00000658-200009000-00007
- Omloo JM, Lagarde SM, Hulscher JB, et al. Extended transthoracic resection compared with limited transhiatal resection for adenocarcinoma of the mid/distal esophagus: five-year survival of a randomized clinical trial. Ann Surg 2007;246:992–1000. [PubMed] DOI:10.1097/SLA.0b013e31815c4037
- Colvin H, Dunning J, Khan OA. Transthoracic versus transhiatal esophagectomy for distal esophageal cancer: which is superior. Interact Cardiovasc Thorac Surg 2011;12:265–8. [PubMed] DOI:10.1510/icvts.2010.252148
- Barbour AP, Rizk NP, Gonen M, et al. Adenocarcinoma of the gastroesophageal junction: influence of esophageal resection margin and operative approach on outcome. Ann Surg 2007;246:1–8. [PubMed] DOI:10.1097/01.sla.0000255563.65157.d2
- Kurokawa Y, Sasako M, Sano T, et al. Ten-year follow-up results of a randomized clinical trial comparing left thoracoabdominal and abdominal transhiatal approaches to total gastrectomy for adenocarcinoma of the oesophagogastric junction or gastric cardia. Br J Surg 2015;102:341–8. [PubMed] DOI:10.1002/bjs.2015.102.issue-4
- Carboni F, Lorusso R, Santoro R, et al. Adenocarcinoma of the esophagogastric junction: the role of abdominal-transhiatal resection. Ann Surg Oncol 2009;16:304–10. [PubMed] DOI:10.1245/s10434-008-0247-x
- Japanese Gastric Cancer Association. Japanese classification of gastric carcinoma: 3rd English edition. Gastric Cancer 2011;14:101–12. [PubMed] DOI:10.1007/s10120-011-0041-5
- Hosokawa Y, Kinoshita T, Konishi M, et al. Clinico-pathological features and prognostic factors of adenocarcinoma of the esophagogastric junction according to Siewert classification: experiences at a single institution in Japan. Ann Surg Oncol 2012;19:677–83. [PubMed] DOI:10.1245/s10434-011-1983-x
- Sasako M, Sano T, Yamamoto S, et al. Left thoraco-abdominal approach versus abdominal-transhiatal approach for gastric cancer of the cardia or subcardia: a randomised controlled trial. Lancet Oncol 2006;7:644–51. [PubMed] DOI:10.1016/S1470-2045(06)70766-5
- Kim JH, Park SS, Kim J, et al. Surgical outcomes for gastric cancer in the upper third of the stomach. World J Surg 2006;30:1870–6. [PubMed] DOI:10.1007/s00268-005-0703-8
- Wen L, Chen XZ, Wu B, et al. Total vs. proximal gastrectomy for proximal gastric cancer: a systematic review and meta-analysis. Hepatogastroenterology 2012;59:633–40. [PubMed]
- Japanese Gastric Cancer Association. Japanese gastric cancer treatment guidelines 2010 (ver. 3). Gastric Cancer 2011;14:113–23. [PubMed] DOI:10.1007/s10120-011-0042-4
- Yamamoto M, Baba H, Egashira A, et al. Adenocarcinoma of the esophagogastric junction in Japan. Hepatogastro-enterology 2008;55:103–7.
- Kim KT, Jeong O, Jung MR, et al. Outcomes of abdominal total gastrectomy for type II and III gastroesophageal junction tumors: single center’s experience in Korea. J Gastric Cancer 2012;12:36–42. [PubMed] DOI:10.5230/jgc.2012.12.1.36
- Hulscher JB, van Sandick JW, de Boer AG, et al. Extended transthoracic resection compared with limited transhiatal resection for adenocarcinoma of the esophagus. N Engl J Med 2002;347:1662–9. [PubMed] DOI:10.1056/NEJMoa022343
- Zhang WH, Chen XZ, Liu K, et al. Comparison of the clinicopathological characteristics and the survival outcomes between the Siewert type II/III adeno-carcinomas. Med Oncol 2014;31:116. [PubMed] DOI:10.1007/s12032-014-0116-3
- Morgagni P, Garcea D, Marrelli D, et al. Resection line involvement after gastric cancer surgery: clinical outcome in nonsurgically retreated patients. World J Surg 2008;32:2661–7. [PubMed] DOI:10.1007/s00268-008-9747-x
- Feith M, Stein HJ, Siewert JR. Adenocarcinoma of the esophagogastric junction: surgical therapy based on 1602 consecutive resected patients. Surg Oncol Clin N Am 2006;15:751–64. [PubMed] DOI:10.1016/j.soc.2006.07.015
- Hasegawa S, Yoshikawa T, Cho H, et al. Is adeno-carcinoma of the esophagogastric junction different between Japan and western countries? The incidence and clinicopathological features at a Japanese high-volume cancer center. World J Surg 2009;33:95–103. [PubMed] DOI:10.1007/s00268-008-9740-4
- Mullen JT, Davenport DL, Hutter MM, et al. Impact of body mass index on perioperative outcomes in patients undergoing major intra-abdominal cancer surgery. Ann Surg Oncol 2008;15:2164–72. [PubMed] DOI:10.1245/s10434-008-9990-2
- Wong JY, Shridhar R, Almhanna K, et al. The impact of body mass index on esophageal cancer. Cancer Control 2013;20:138–43. [PubMed]
- Zhang SS, Yang H, Luo KJ, et al. The impact of body mass index on complication and survival in resected oesophageal cancer: a clinical-based cohort and meta-analysis. Br J Cancer 2013;109:2894–903. [PubMed] DOI:10.1038/bjc.2013.666
