Survival difference between EGFR Del19 and L858R mutant advanced non-small cell lung cancer patients receiving gefitinib: a propensity score matching analysis
Introduction
Lung cancer imposes a major disease burden on the world. It remains the most commonly diagnosed cancer and the greatest cause of cancer-related death (1). In non-small-cell lung cancer (NSCLC), epidermal growth factor receptor (EGFR) exon 19 deletion (Del19) and exon 21 Leu858Arg substitution (L858R), which account for 90% of all EGFR mutations, are associated with favorable outcomes in patients treated with EGFR-tyrosine kinase inhibitors (TKIs) (2-5). Many retrospective and prospective studies, as well as meta-analyses, that have studied NSCLC patients with various lines of EGFR-TKI treatment have demonstrated longer progression-free survival (PFS) and occasionally more favorable overall survival (OS) in those with Del19 than in those with the L858R or other mutations (6-8). In contrast, other clinical studies, including phase III trials, have demonstrated no difference in the efficacy of EGFR-TKI treatment according to the EGFR mutation type (9-12). Therefore, whether there are differences in survival between patients with these common EGFR mutations remains controversial. In this study, we analyzed the clinical benefit of gefitinib in advanced NSCLC, and explored the survival differences between two common mutation subtypes.
Materials and methods
Patients
Our single-center, retrospective study was approved by the Ethics Committee of the Peking University Cancer Hospital in Beijing, China. Inclusion criteria were patients who were diagnosed with advanced NSCLC between September 2006 and September 2016 and who received gefitinib treatment. Exclusion criteria were patients under 18 years old or for whom results for EGFR mutation type were unavailable. A total of 204 patients were enrolled in our study. Data, such as age, sex, histological type, clinical stage, brain metastases and treatment regimens, were collected from hospital computer information systems and telephone calls for analysis.
Evaluation
We used the amplification refractory mutation system-polymerase chain reaction (ARMS-PCR) method to examine mutations in tumor tissues. The diagnosis of brain metastasis was judged by an independent radiologist based on brain magnetic resonance imaging (MRI) findings. Disease progression was assessed according to Response Evaluation Criteria In Solid Tumors (RECIST) version 1.1 (13). All staging procedures were carried out using the 7th Union for International Cancer Control tumor node metastasis (TNM) classification. The primary endpoint of our study was PFS, which was defined as the duration from the onset of gefitinib treatment to disease progression (local, regional, and/or distant) or death from any cause. The secondary endpoint of our study was OS. The last follow-up was on May 17, 2017. Data were censored on the last contact date. Patients who were still alive at the final follow-up were regarded as censored, and the duration between the initial treatment and the final follow-up was included in the survival analysis.
Statistical analysis
Patients were categorized into two groups based on EGFR mutation subtype: the Del19 group and the L858R group. Categorical data are reported as number with percentage, and continuous data are reported as either x±s or median (interquartile range), as appropriate. Categorical data were compared using the Chi-square test or Fisher’s exact test. PFS and OS were estimated using the Kaplan-Meier method. Comparison of PFS and OS between groups was performed using the log-rank test. Additionally, we conducted unadjusted and multivariable Cox proportional hazards models controlling for all demographic and clinical characteristics to examine whether the mutation type had an impact on PFS or OS among our participants. Groups selected for subgroup analyses were selected a priori, including subgroups that were based on different therapy lines.
To ensure that the treatment groups were as similar as possible, we used propensity score matching (PSM). First, logistic regression was performed to estimate the probability of mutation subtype as a function of the following characteristics: age, sex, histological type, clinical stage, brain metastases, mutation frequency and therapy line. Next, matching (1:1) on the propensity score was performed using a nearest neighbor-matching algorithm, with a maximum caliper distance of 0.25 of the standard deviation of the propensity score. To assess the performance of the matching, baseline categorical variables were compared between the matched groups, and standardized differences were calculated for which a difference between –0.1 and 0.1 is generally considered negligible.
Statistical analysis was performed with R software (Version 3.3.3; R Foundation for Statistical Computing, Vienna, Austria). The reported significance levels were all two-sided, with statistical significance set at 0.05.
Results
Clinical characteristics
Clinical characteristics and survival data for the 204 patients enrolled in this retrospective study were summarized and analyzed. The median patient age was 59 (range: 20–90) years. There were 68 male patients and 136 female patients, suggesting that EGFR mutations are more often present in female patients. There were 194 adenocarcinomas and 10 non-adenocarcinomas, indicating the predominant presence of EGFR mutations in adenocarcinomas. Common adverse effects at any grade included acneiform rash (54.6%), diarrhea (10.1%), and transaminase elevation (7.8%). No patients discontinued gefitinib due to treatment-related toxicity.
Using the primary data, we compared the age, sex, histology, brain metastases and therapy line type among different mutation types. In the Del19 group, 21.4% patients were aged 65 years or above, whereas this proportion was significantly higher in the L858R group (38.4%, P=0.015). No significant differences were seen for sex, histology, or other baseline characteristics (Table 1).
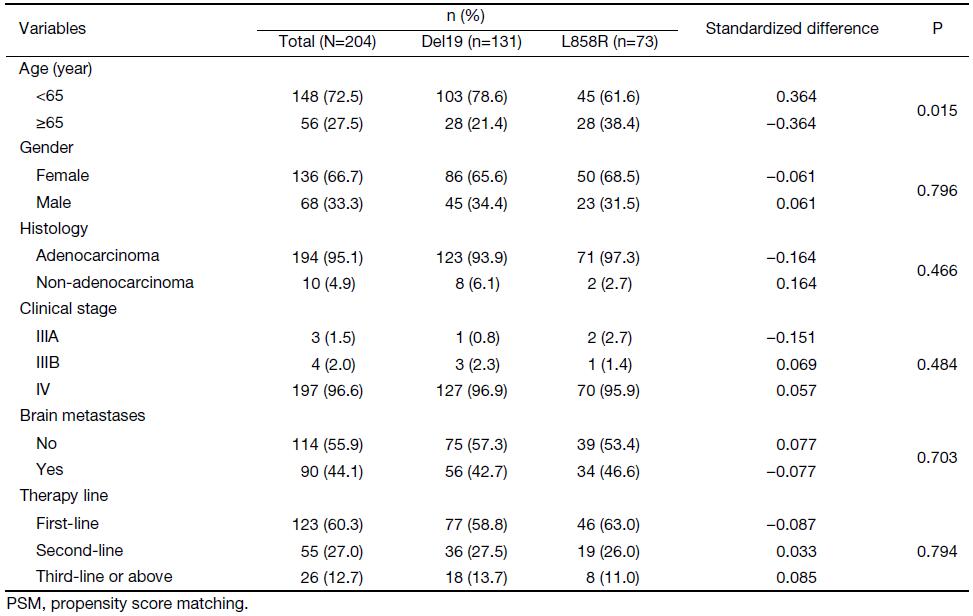
Full table
Survival comparison and PSM
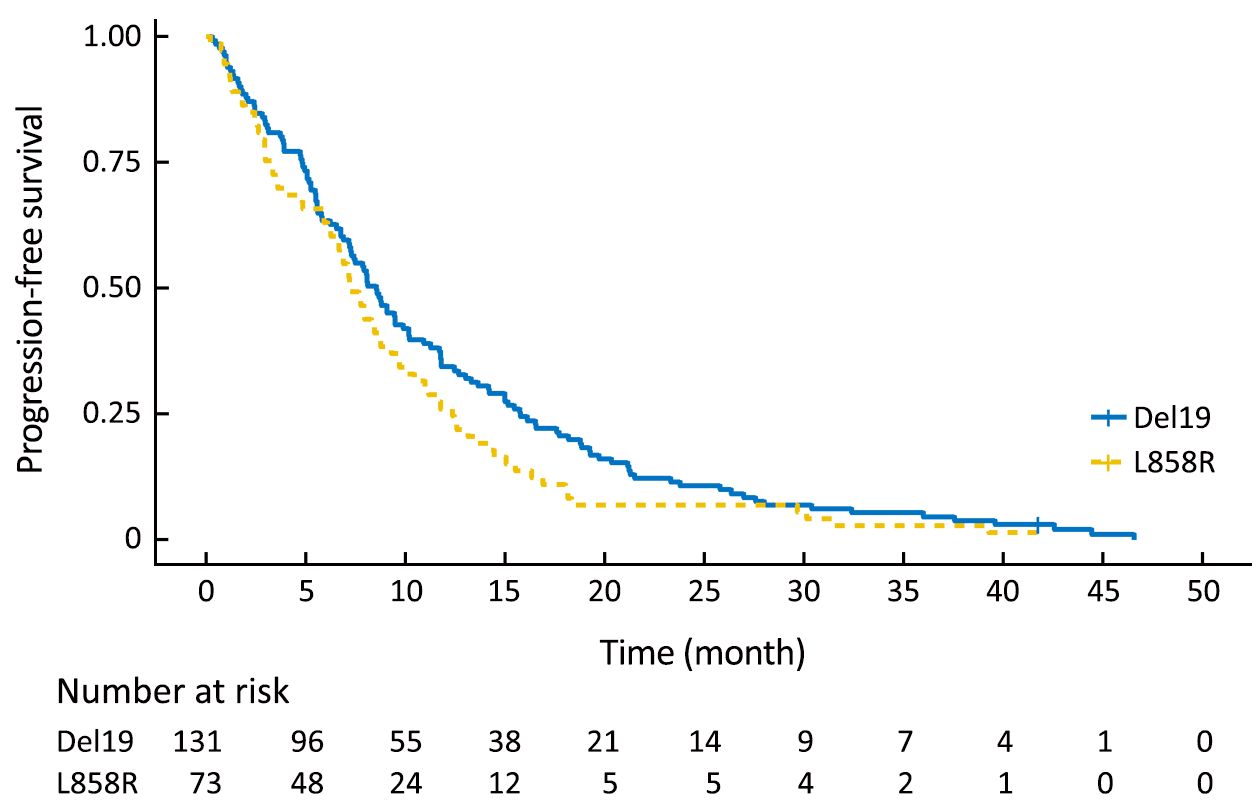
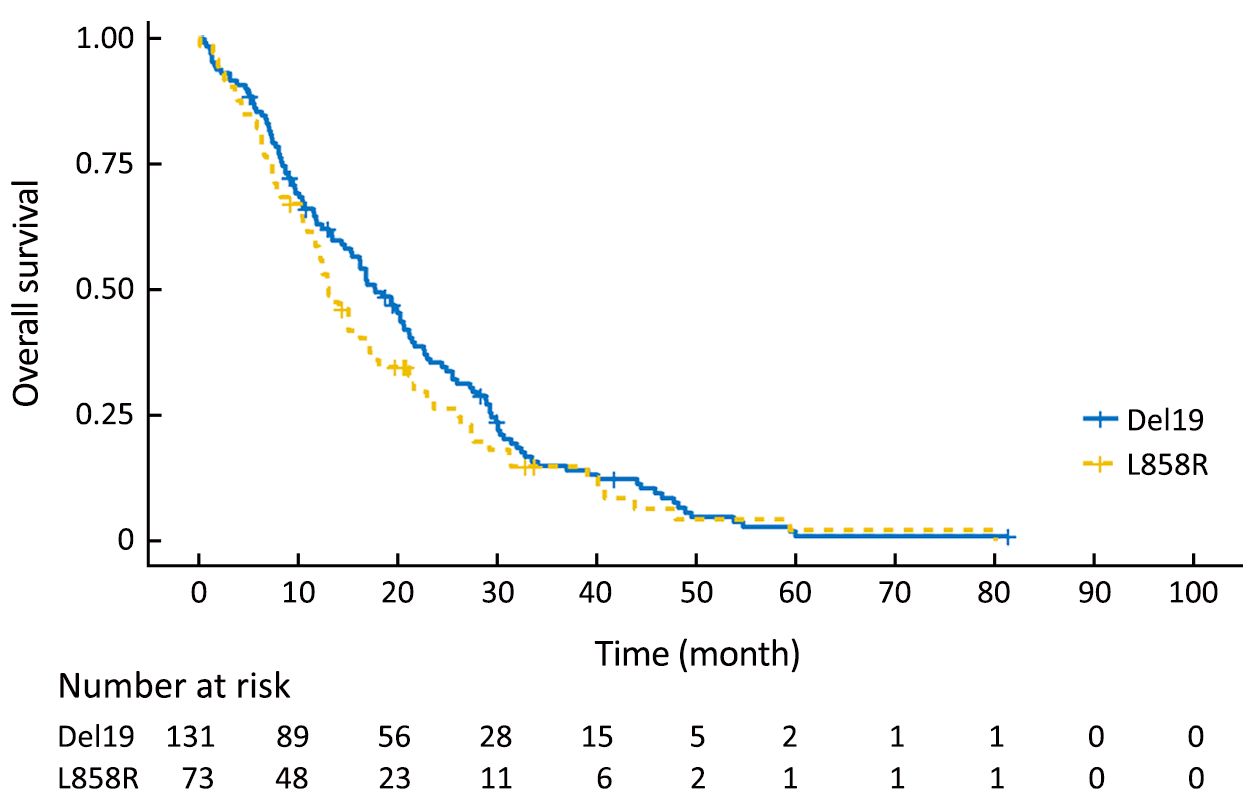
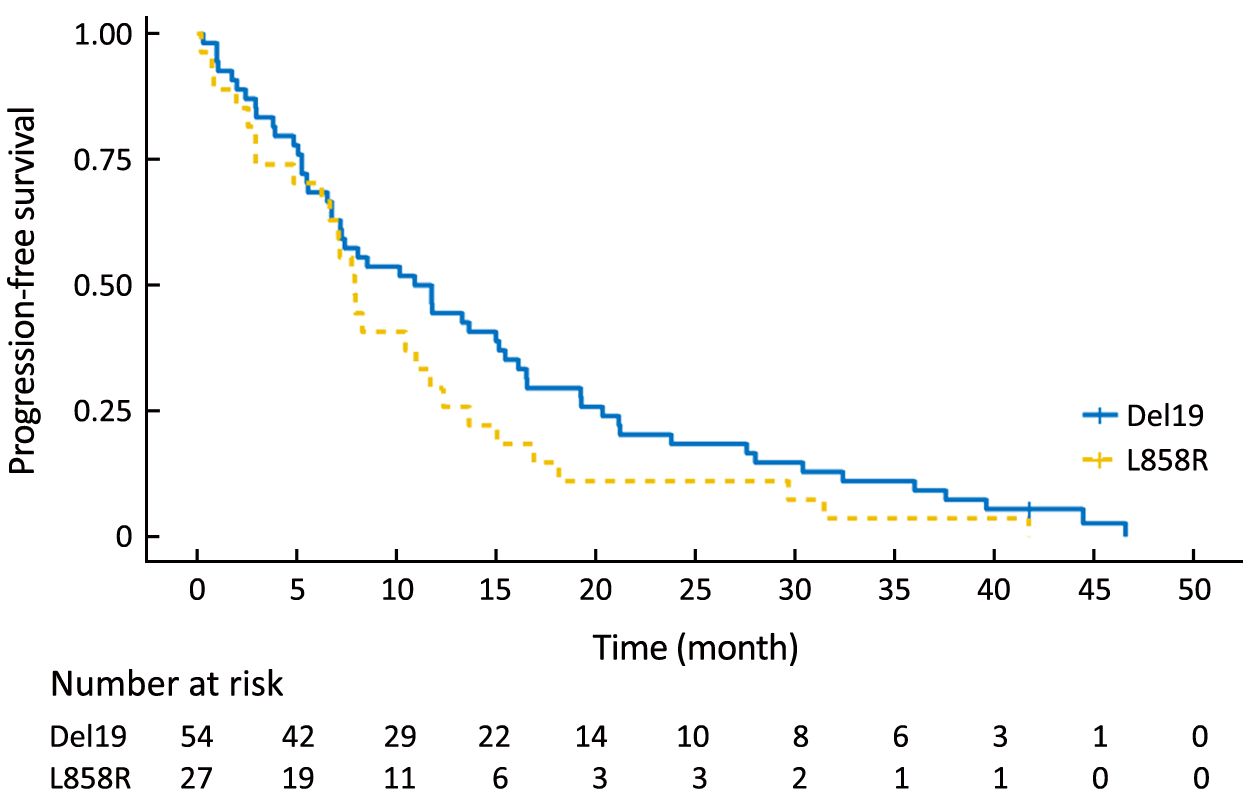
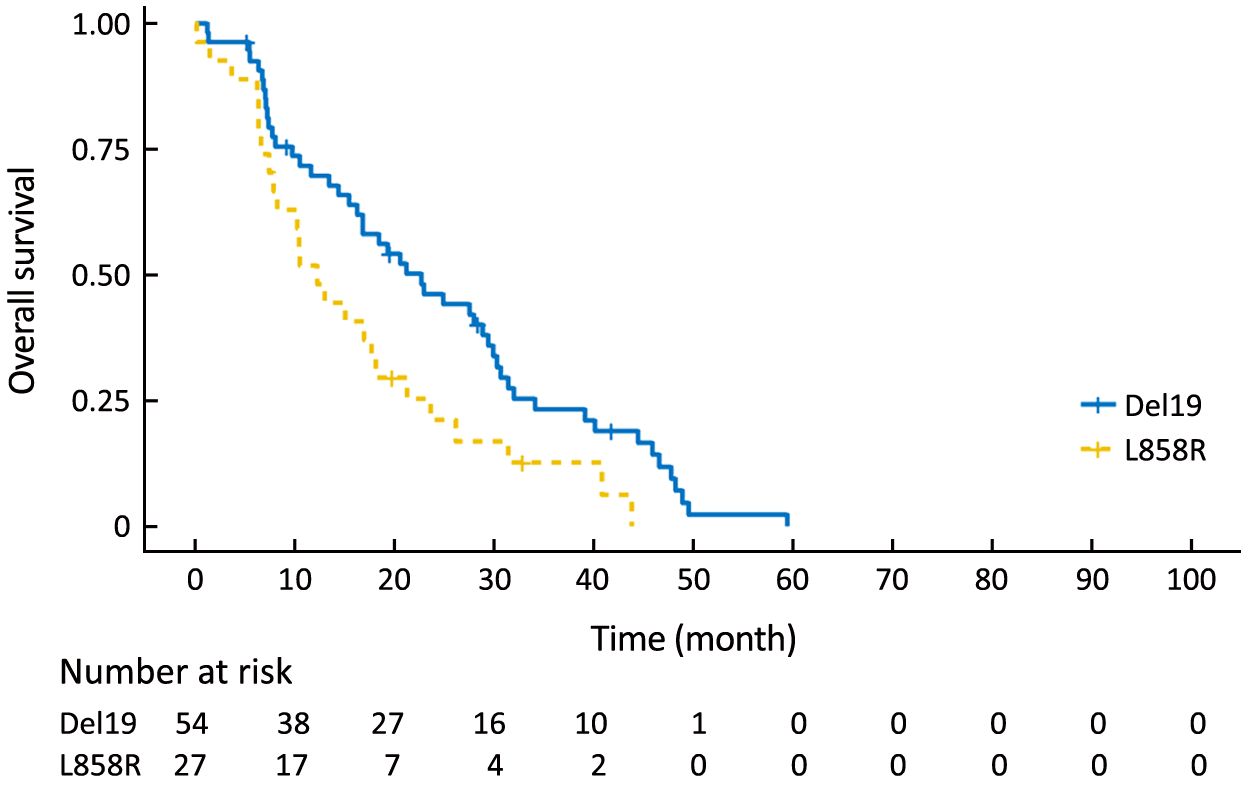
Using the primary data, we analyzed survival in the two groups. The PFS of the Del19 group tended to be longer than that of the L858R group, but did not reach statistical significance [8.6 vs. 7.2 months, respectively; hazard ratio (HR): 1.304; 95% confidence interval (95% CI): 0.976–1.742; P=0.072] (Figure 1). The OS of the Del19 group also tended to be longer than that of the L858R group, but did not reach statistical significance (17.8 vs. 13.1 months, respectively; HR: 1.192; 95% CI: 0.882–1.611; P=0.253) (Figure 2).
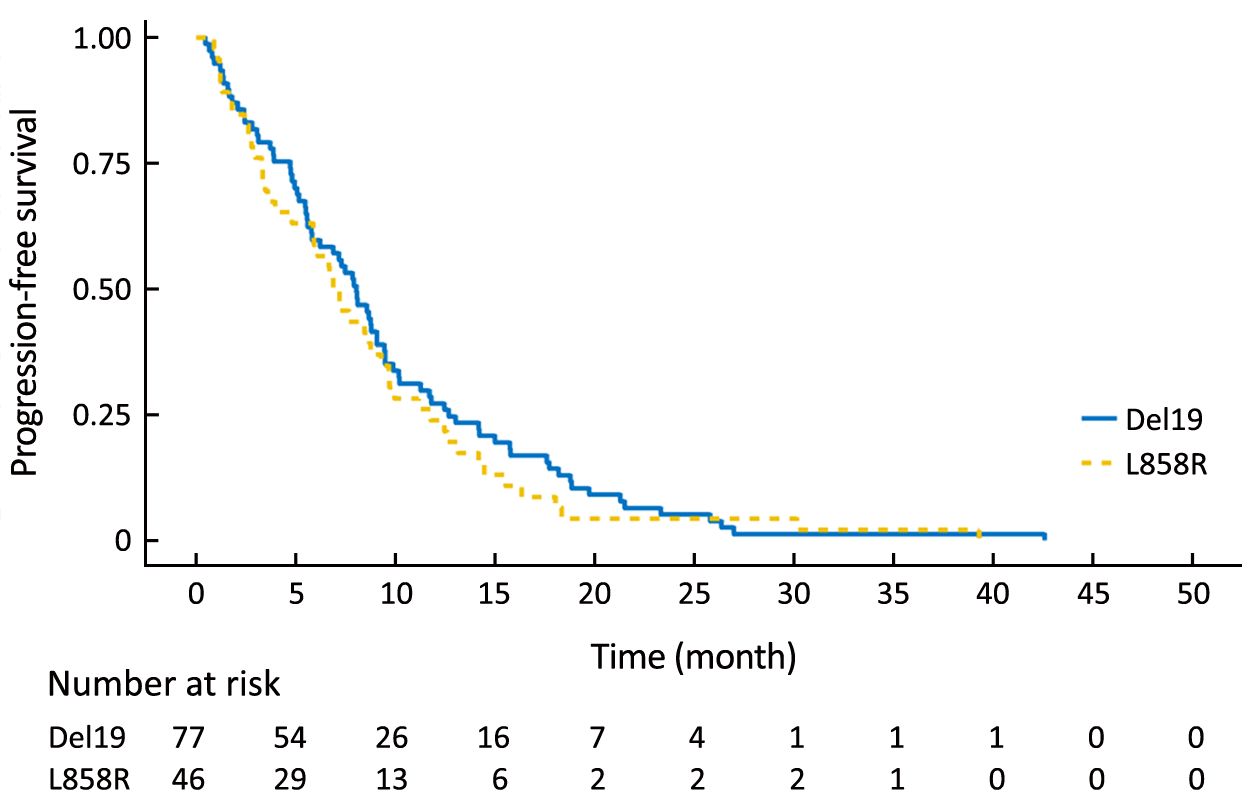
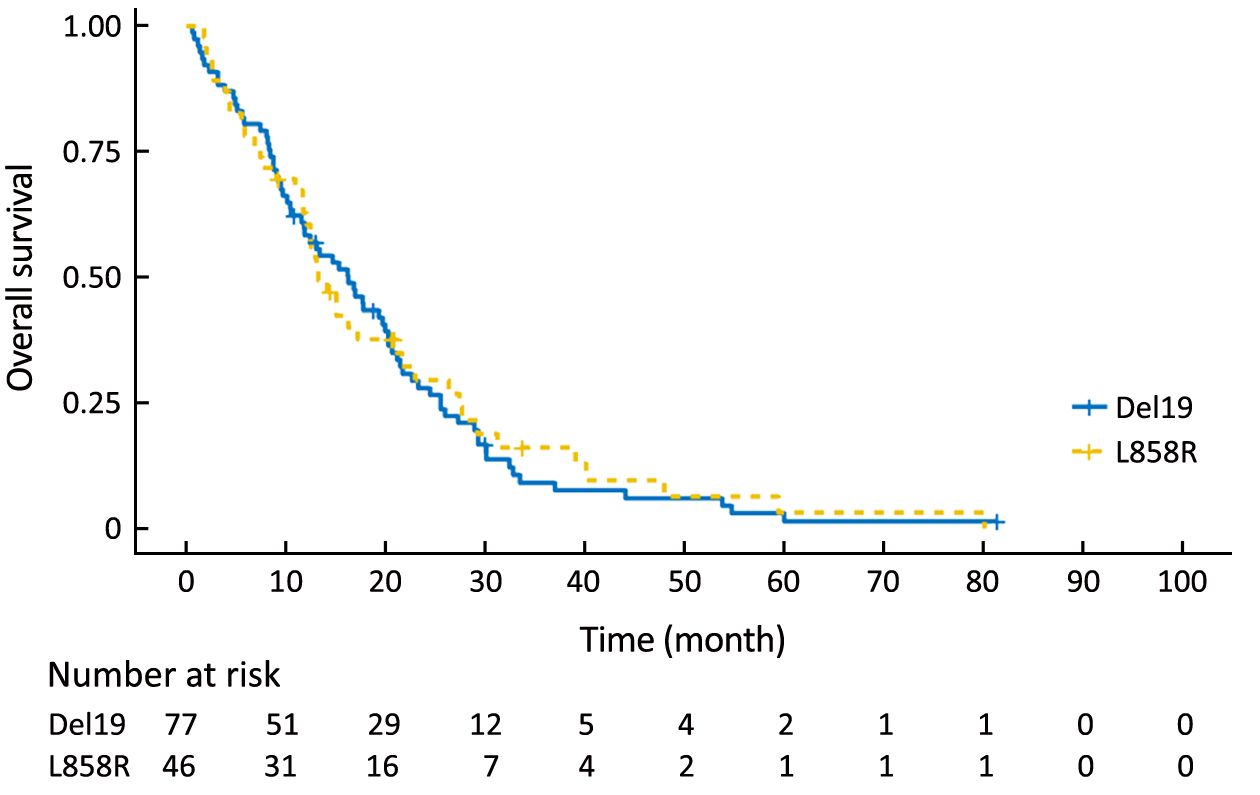
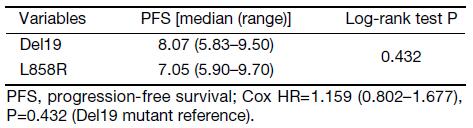
To eliminate potential imbalances between the two groups, we performed PSM. After the matching, the Del19 and L858R groups were well balanced for various factors, including age (Table 2). Following PSM, the differences in PFS and OS between the two groups were reduced. The PFS of the Del19 group was similar to that of the L858R group (7.3 vs. 7.2 months, respectively; HR: 1.287; 95% CI: 0.909–1.824; P=0.155) (Figure 3). No significant difference was detected in OS between the Del19 and L858R groups (16.9 vs. 13.1 months, respectively; HR: 1.187; 95% CI: 0.835–1.688; P=0.339) (Figure 4).
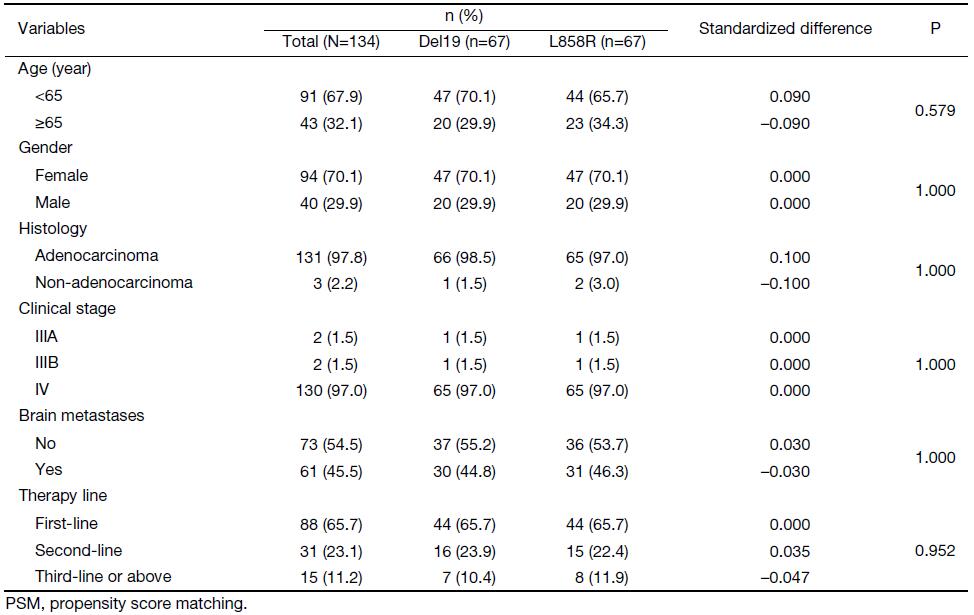
Full table
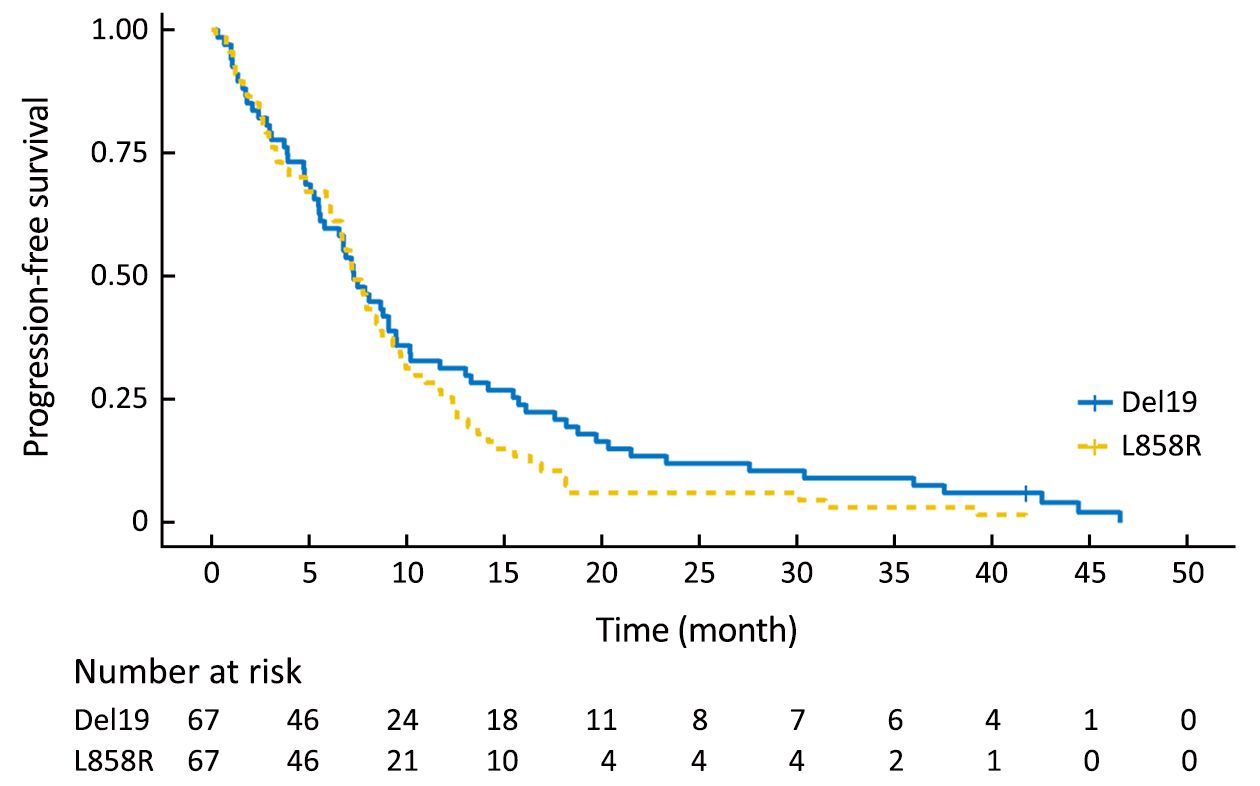
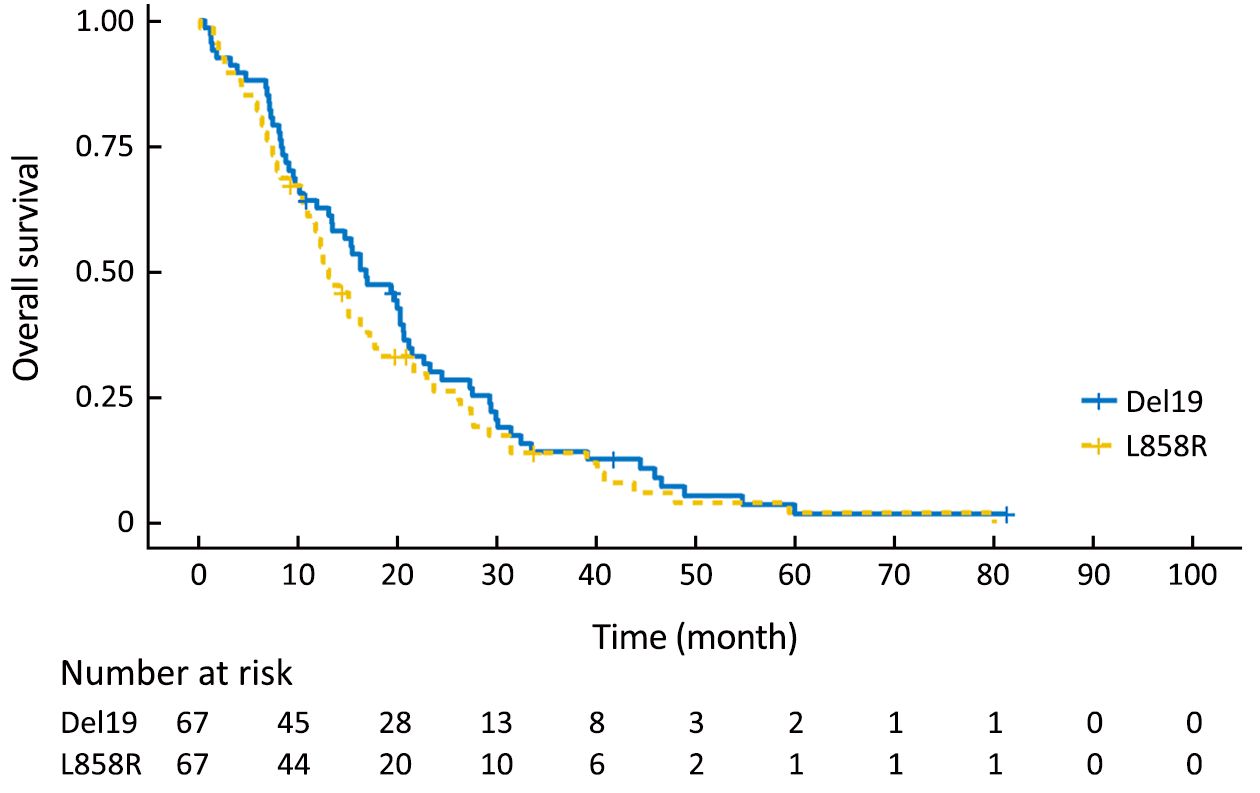
To confirm the results of the PSM analysis, we next performed a sensitivity analysis based on a multivariate Cox proportional hazards regression model in the pre-matching cohort. Our data showed results similar to the PSM analysis: mutation type did not reach statistical significance for either PFS (HR: 1.323; 95% CI: 0.966–1.811; P=0.080) or OS (HR: 1.244; 95% CI: 0.906–1.710; P=0.177).
Survival analysis for different therapy lines
There were 77 and 46 patients who received first-line gefitinib therapy in the Del19 group and L858R groups, respectively, while 54 and 27 patients received gefitinib as a second-line therapy or above in the Del19 group and L858R groups, respectively. The disease control rate was 87.1% in the first-line setting, and 66.1% in the second-line setting.
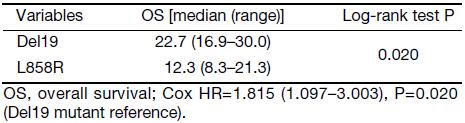
We compared the PFS and OS between the Del19 and L858R groups within different therapy lines. The OS of the Del19 group was significantly longer than that of the L858R group in the second-line or further management setting (22.7 vs. 12.3 months, respectively; P=0.020), but not in the first-line setting (Supplementary data). The stratification of different therapy lines was not performed in the PSM due to the limited sample size.
Discussion
Although there were some reports showing that EGFR-TKI treatment was more effective in the Del19 group than in the L858R group, results on the survival differences between Del19 and L858R mutation subtypes remained inconsistent (14,15). In this study, we investigated the characteristics and survival outcomes of Del19 and L858R mutation patients receiving gefitinib. In contrast to prior studies, we performed rigorous adjustment for baseline differences by PSM to overcome the impact of bias and potential confounding factors in this retrospective study. After PSM, the 67 patients in the Del19 group and the 67 patients in the L858R group were well matched. This ensured that our study conclusion was more solid and convincing.
In keeping with previous reports, we found that these mutations were predominantly present in female patients, and in the adenocarcinoma histotype (6,8,16). However, the OS in our study was shorter in contrast with the data obtained by Ke et al. (16.3 vs. 33.3 in the Del19 group; 13.3 vs. 26.4 months in the L858R group) (17). We hypothesized that reasons for such differences might include the fact that there was more brain metastasis in our study (44.1%), which could lead to shorter OS.
In this study, the PFS and OS of the Del19 group tended to be longer than those of L858R group, which is in keeping with previous studies. Choi et al. reported that patients with Del19 (n=28) had a significantly longer median PFS (20 vs. 8 months, P=0.004) compared with those with L858R mutation (n=25) and uncommon or dual mutations (n=7) (18). For OS, Kim et al. reported that OS was significantly better in patients with Del19 than those with L858R (24-month OS rate was 72.1% vs. 32.0%, P=0.0148) (19). Koyama et al. reported that a significant improvement in OS was observed in the group harboring Del19 compared with those harboring L858R (NR vs. 839 days, respectively; HR: 0.374; P=0.024) (20). Ke et al. recently reported the median OS was 33.3 (95% CI: 28.9–37.7) months in patients with EGFR Del19, and 26.4 (95% CI: 23.2–29.6) months in those with the L858R mutation (17). However, no significant difference was seen in the PFS or OS between the two groups in our study. Possible reasons for this could include: 1) this was a single-center, retrospective study with certain limitations; 2) the study was based on a Chinese population, which may have resulted in different results compared to those from other nations; and 3) the post-TKI treatment may have influenced OS.
In this study, we found that the Del19 group was significantly younger than the L858R group in age. Similar findings have been previously reported. For instance, it has been shown that the percentage of patients aged <55 years was significantly higher in the Del19 group than in the L858R group (47.5% vs. 31.8%, respectively; P=0.021) (17). Zheng et al. also showed that the Del19 group was younger than the L858R group in age (21). When we used PSM to exclude such differences, the survival gap between the Del19 and L858R groups was reduced. We propose that the age difference could also contribute to the survival differences between the Del19 and L858R groups, which need to be explored in future comparative analysis.
We found a significant difference in the OS between the Del19 and L858R groups in second-line and further settings but not in a first-line setting. This is in agreement with the findings of a previous report (22). PSM analysis was not employed in this part because it would result in a sample size reduction that would limit any conclusions that could be drawn. Our result indicates that the lines of EGFR-TKI therapy should be discriminated in future studies when OS was set as the primary endpoint.
This study has some limitations. Firstly, the sample size was relatively small. Secondly, we did not explore the resistance mechanism for Del19 and L858R mutations. It has been shown that the Del19 mutation was more often associated with the EGFR exon 20 T790M mutation, which could be the reason for longer PFS and OS in patients with the Del19 mutation compared to those with the L858R mutation (17). Further research in this field is necessary.
Conclusions
We compared the PFS and OS between the Del19 and L858R mutation groups. Before the PSM, the PFS and OS of the Del19 group tended to be longer than that of the L858R group, but did not reach statistical significance. After PSM, the survival gap between the Del19 and L858R groups was reduced, and no significant differences were detected in the PFS or OS between the two groups. We found significant differences in age between the Del19 and L858R groups, which might contribute to the survival differences. Prognostic controlled clinical trials are warranted to determine the potential survival difference between the patients with Del19 and L858R mutations receiving gefitinib.
Acknowledgements
This work was supported by the Beijing Natural Science Foundation (Commission No. 7162038) and the Beijing Municipal Administration of Hospitals’ Youth Program (Commission No. QML20161101).
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Supplement data
Patients were classified into the first-line setting or the second-line and beyond setting, according to the line of their epidermal growth factor receptor tyrosine kinase inhibitors (EGFR-TKI) treatment. The progression-free survival (PFS) and overall survival (OS) were compared between Del19 and L858R groups.
In the first-line setting, no significant difference was seen in the PFS or OS between Del19 and L858R groups (PFS: Supplementary Table S1, Supplementary Figure S1; OS: Supplementary Table S2, Supplementary Figure S2).
In the second-line and beyond setting, the PFS of Del19 group tended to be longer than that of L858R group, but did not reach statistical significance (Supplementary Table S3, Supplementary Figure S3). The OS of Del19 group was significantly longer than that of L858R group (Supplementary Table S4, Supplementary Figure S4).
Full table

Full table

Full table
Full table
References
- Cheng TY, Cramb SM, Baade PD, et al. The International Epidemiology of Lung Cancer: Latest Trends, Disparities, and Tumor Characteristics. J Thorac Oncol 2016;11:1653–71. DOI:10.1016/j.jtho.2016.05.021
- Masters GA, Temin S, Azzoli CG, et al. Systemic Therapy for Stage IV Non-Small-Cell Lung Cancer: American Society of Clinical Oncology Clinical Practice Guideline Update. J Clin Oncol 2015;33:3488–515. DOI:10.1200/JCO.2015.62.1342
- Steuer CE, Ramalingam SS. Targeting EGFR in lung cancer: lessons learned and future perspectives. Mol Aspects Med 2015;45:67–73. DOI:10.1016/j.mam.2015.05.004
- Maemondo M, Inoue A, Kobayashi K, et al. Gefitinib or chemotherapy for non-small-cell lung cancer with mutated EGFR. N Engl J Med 2010;362:2380–8. DOI:10.1056/NEJMoa0909530
- Li L, Zhang Z, Bie Z, et al. Epidermal growth factor receptor mutation analysis in cytological specimens and responsiveness to gefitinib in advanced non-small cell lung cancer patients. Chin J Cancer Res 2015;27:294–300. DOI:10.3978/j.issn.1000-9604.2015.05.03
- Zhang Y, Sheng J, Kang S, et al. Patients with exon 19 deletion were associated with longer progression-free survival compared to those with L858R mutation after first-line EGFR-TKIs for advanced non-small cell lung cancer: a meta-analysis. PLoS One 2014;9:e107161. DOI:10.1371/journal.pone.0107161
- Yang JC, Wu YL, Schuler M, et al. Afatinib versus cisplatin-based chemotherapy for EGFR mutation-positive lung adenocarcinoma (LUX-Lung 3 and LUX-Lung 6): analysis of overall survival data from two randomised, phase 3 trials. Lancet Oncol 2015;16:141–51. DOI:10.1016/S1470-2045(14)71173-8
- Lee CK, Wu YL, Ding PN, et al. Impact of specific epidermal growth factor receptor (EGFR) mutations and clinical characteristics on outcomes after treatment with EGFR tyrosine kinase inhibitors versus chemotherapy in EGFR-mutant lung cancer: A Meta-analysis. J Clin Oncol 2015;33:1958–65. DOI:10.1200/JCO.2014.58.1736
- Xue ZX, Wen WX, Zhuang Y, et al. Comparison of the efficacy of icotinib in patients with non-small-cell lung cancer according to the type of epidermal growth factor receptor mutation. Mol Clin Oncol 2016;5:265–68. DOI:10.3892/mco.2016.956
- Shen YW, Zhang XM, Li ST, et al. Efficacy and safety of icotinib as first-line therapy in patients with advanced non-small-cell lung cancer. Onco Targets Ther 2016;9:929–35. DOI:10.2147/OTT.S98363
- Li X, Qin N, Wang J, et al. Clinical observation of icotinib hydrochloride for advanced non-small cell lung cancer patients with EGFR status identified. Zhongguo Fei Ai Za Zhi (in Chinese) 2015;18:734–9. DOI:10.3779/j.issn.1009-3419.2015.12.04
- Deng W, Lei Y, Liu S, et al. Comparing overall survival between first generation EGFR-TKIs and chemotherapy in lung cancer patients with Del19/L858R. Chin J Cancer Res 2016;28:339–47. DOI:10.21147/j.issn.1000-9604.2016.03.08
- Eisenhauer EA, Therasse P, Bogaerts J, et al. New response evaluation criteria in solid tumours: Revised RECIST guideline (version 1.1). Eur J Cancer 2009;45:228–47. DOI:10.1016/j.ejca.2008.10.026
- Zhang Y, Wang Z, Hao X, et al. Clinical characteristics and response to tyrosine kinase inhibitors of patients with non-small cell lung cancer harboring uncommon epidermal growth factor receptor mutations. Chin J Cancer Res 2017;29:18–24. DOI:10.21147/j.issn.1000-9604.2017.01.03
- Ellis PM, Coakley N, Feld R, et al. Use of the epidermal growth factor receptor inhibitors gefitinib, erlotinib, afatinib, dacomitinib, and icotinib in the treatment of non-small-cell lung cancer: a systematic review. Curr Oncol 2015;22:e183–215. DOI:10.3747/co.22.2566
- Lee VH, Leung DK, Choy TS, et al. Efficacy and safety of afatinib in Chinese patients with EGFR-mutated metastatic non-small-cell lung cancer (NSCLC) previously responsive to first-generation tyrosine-kinase inhibitors (TKI) and chemotherapy: comparison with historical cohort using erlotinib. BMC Cancer 2016;16:147. DOI:10.1186/s12885-016-2201-9
- Ke EE, Zhou Q, Zhang QY, et al. A higher proportion of the EGFR T790M mutation may contribute to the better survival of patients with exon 19 deletions compared with those with L858R. J Thorac Oncol 2017;12:1368–75. DOI:10.1016/j.jtho.2017.05.018
- Choi YW, Jeon SY, Jeong GS, et al. EGFR exon 19 deletion is associated with favorable overall survival after first-line geftinib therapy in advanced non-small cell lung cancer patients. Am J Clin Oncol 2016. [Epub ahead of print].
- Kim DW, Lee SH, Lee JS, et al. A multicenter phase II study to evaluate the efficacy and safety of gefitinib as first-line treatment for Korean patients with advanced pulmonary adenocarcinoma harboring EGFR mutations. Lung Cancer 2011;71:65–9. DOI:10.1016/j.lungcan.2010.04.005
- Koyama N, Watanabe Y, Iwai Y, et al. Distinct benefit of overall survival between patients with non-small-cell lung cancer harboring EGFR exon 19 deletion and Exon 21 L858R substitution. Chemotherapy 2017;62:151–8. DOI:10.1159/000454944
- Zheng Z, Jin X, Lin B, et al. Efficacy of second-line tyrosine kinase inhibitors in the treatment of metastatic advanced non-small-cell lung cancer harboring exon 19 and 21 EGFR mutations. J Cancer 2017;8:597–605. DOI:10.7150/jca.16959
- Zheng Z, Xie D, Su H, et al. Treatment outcome comparisons between exons 19 and 21 EGFR mutations for non-small-cell lung cancer patients with malignant pleural effusion after first-line and second-line tyrosine kinase inhibitors. Tumour Biol 2017;39:1010428317706211. DOI:10.1177/1010428317706211
