Retrospective analysis of interventional treatment of hepatic metastasis from gastroenteropancreatic neuroendocrine tumors
Introduction
Derived from peptidergic neuron and neuroendocrine cells, neuroendocrine tumors (NETs) are a group of heterogeneous tumors. Gastroenteropancreatic neuroendocrine tumors (GEP-NETs) account for 55%–70% of NETs (1,2). Originated from neuroendocrine system, GEP-NETs are found in non-specific organs. Functional GEP-NETs secrete a variety of hormones that can trigger symptoms. Most GEP-NETs are nonfunctional, and its clinical manifestations only occurred lately in the process of disease. The incidence and prevalence of GEP-NETs have increased approximately 5-fold to 5.25/100 thousand in the past 30 years (3,4). Now, GEP-NETs rank the second common gastrointestinal tumor of the world, and many factors are related to the survival of GEP-NETs (5).
Surgical resection is feasible to treat NETs and metastasis, which will increase survival in some patients for its effect and efficiency. Unfortunately, only 5%–10% of diagnosed patients are eligible for resection with a risk of extremely high recurrence rates. It was reported that 94% of patients will relapse within 5 years (6,7). Metastasis usually occurs in the prophase of NETs. The liver is the most common site of metastasis, accounting for 10%–65% (8,9). Liver metastasis has occurred in most patients at the time of diagnosis. Unresectable liver metastasis still should be treated actively to palliate symptoms and slow down or stabilize tumor growth. Theoretical studies have shown that hepatic metastases, especially larger metastases, are mainly supplied by the hepatic artery (10,11), which provided a theoretical basis for the interventional treatment of hepatic metastases derived from NETs.
Materials and methods
This study retrospectively reviewed the interventional treatment of patients with GEP-NETs in the Department of Interventional Therapy, Peking University Cancer Hospital, who mainly presented the symptom of hepatic metastasis. This retrospective analysis of the data was approved by the Institutional Review Board of Peking University Cancer Hospital.
Patients
All the patients were pathologically diagnosed based on hepatic aspiration biopsy. Patients received at least one complete cycle of interventional therapy for hepatic metastases. The first cycle of interventional therapy was completed between January 2012 and December 2016. The follow-up period ended in June 2017. All of the cases were followed-up for more than 6 months.
Interventional technique
Patients received transarterial embolization (TAE) in the hepatic tumor, and some patients (pathological grade G3 mostly) combined hepatic arterial infusion chemotherapy (HAIC).
Femoral arterial puncture was achieved with a modified Seldinger method. Angiographies of the celiac artery, hepatic artery and tumor blood vessels were used to determine the location and blood supply of the tumor via a catheter and the microvasculature, respectively. Angiographies of the superior mesenteric artery, diaphragm artery, or middle adrenal artery were added when necessary. The catheters were super-selected into the liver and tumor blood vessels. Granular embolic agent and Lipiodol ultrafluid were chosen for embolization. Some patients were treated with HAIC after TAE. The HAIC procedure was performed as following: the catheter was placed in the blood supply artery of the metastasis lesion. Continuous infusion of chemotherapeutic drugs was performed by an external arterial infusion pump via the arterial catheter. Chemotherapy regimen 1 consisted of pumped oxaliplatin 75–100 mg/m2 for 6 h followed by 5-fluorouracil 1.5 g/m2 from 6 to 48 h. Chemotherapy regimen 2 consisted of paclitaxel 135 mg/m2 (docetaxel 75 mg/m2) for 6 h via an arterial pump followed by cisplatin 75 mg/m2 for next 6 h.
Evaluation of interventional treatment
Evaluation of interventional treatment was based on the modified Response Evaluation Criteria in Solid Tumors (mRECIST). The main observed indexes were hepatic progression-free survival (HPFS) and overall survival (OS).
Statistical software SPSS 13.0 (SPSS Inc., Chicago, IL, USA) was used for statistical analysis. Statistical descriptions of categorical data were reported as the frequency (%). HPFS were estimated with the Kaplan-Meier method. Kaplan-Meier curves were plotted by using the log-rank test to demonstrate the associations between survivals and factors. Angiography (hypovascularity/hypervascularity), collateral blood supply or portal vein invasion (yes/no), interventional therapy (TAE/TAE+HAIC) and frequency of intervention (n≤3/n>3) were viewed as general factors in multivariable Cox regression models, which were used to estimate the hazard ratios (HRs) and identify the independent predictors of HPFS. P<0.05 (two-sided) was considered as the cut-off for significance.
Results
Origin of tumor
From January 2012 to December 2016, 50 GEP-NETs patients (22 male, 28 female) were treated in the Department of Interventional Therapy, Peking University Cancer Hospital. Age of patients ranged from 30 to 80 years, with a median age of 54 years. The origin of the tumor included pancreas (n=20), colorectum (n=11), stomach (n=8), small intestine and duodenum (n=7), and liver and gallbladder (n=4).
Angiography appearance of liver metastasis
Tumor vascularity was observed in all cases after detailed angiography. The lesion staining should be judged by consensus of at least 2 experienced interventional physicians. If there is any difference, another chief physician should be judged as the final result.
Only 15 cases were found with poor blood supply, whereas 35 cases with abundant vasculature. Of the 50 patients, 22 had collateral blood supply, portal vein involvement or hepatic arterioportal fistulas. All the patients received a total of 179 times of intervention with a median of 3 times. Of them, 23 patients received embolization alone.
Therapeutic effect
Therapeutic effect was observed and evaluated after a complete cycle, and some patients got good results (Figure 1). Complete remission (CR) was found in 1 case, partial remission (PR) in 18 cases, stable disease (SD) in 27 cases and progressive disease (PD) in 4 cases. The best curative effect during treatment was 4 cases of CR, 28 cases of PR and 18 cases of SD. The valid control rate was 64%.
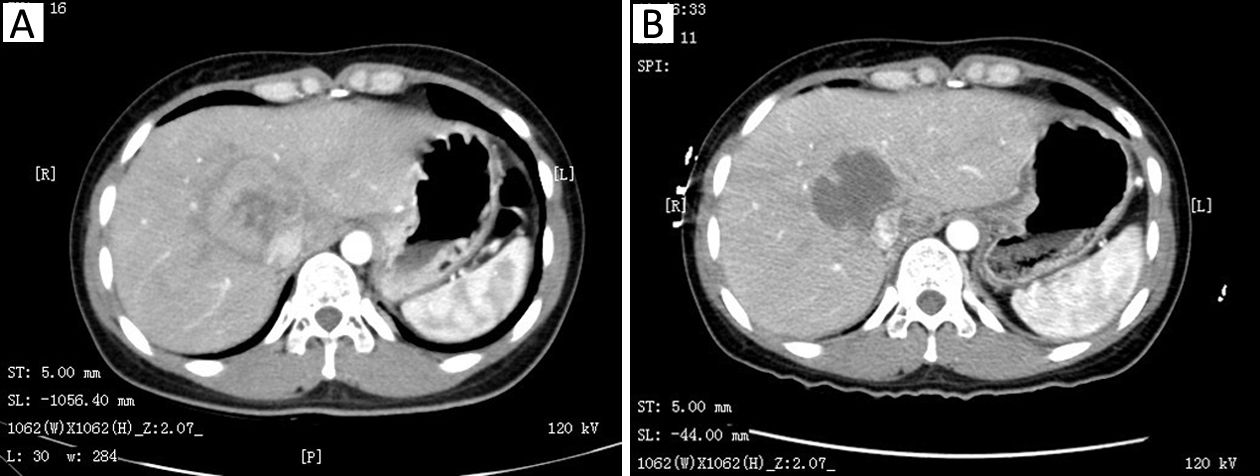
Survival data and response rate
After interventional treatment, the HPFS of all of the patients ranged from 1 to 37 months, with a median of 9 months and a mean of 11 months. In addition, 17 patients died during treatment. The OS ranged from 5 to 36 months, with a mean of 21 months and a median of 19 months.
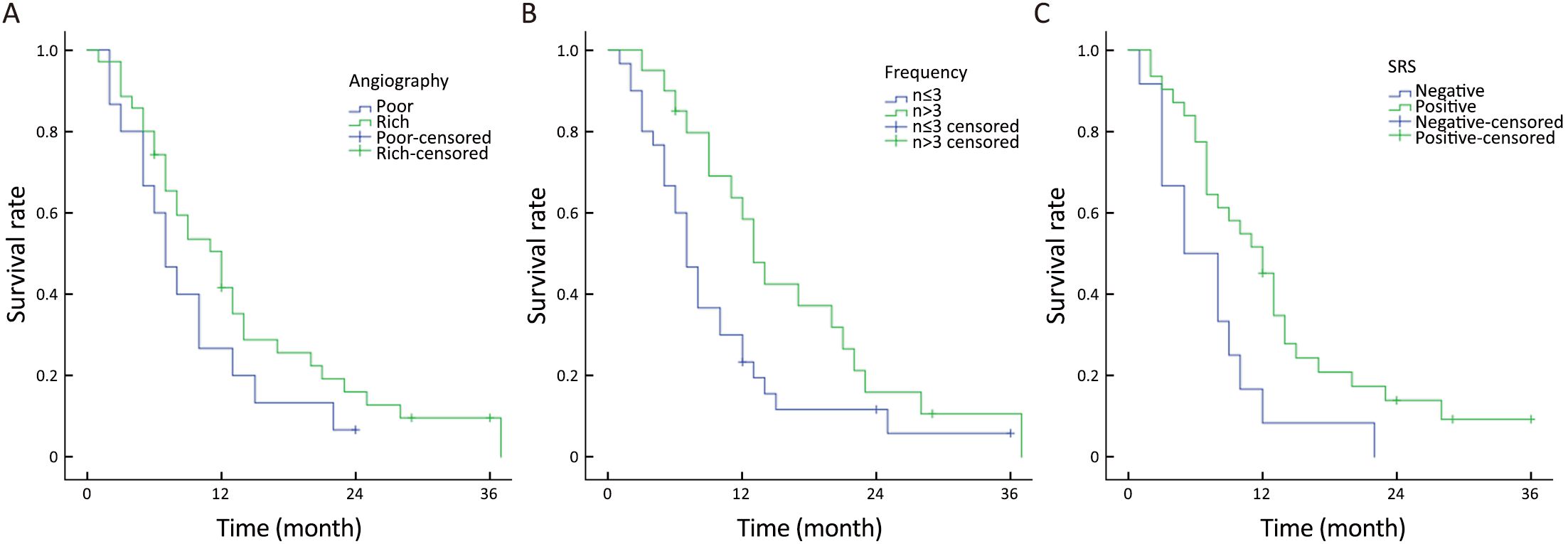
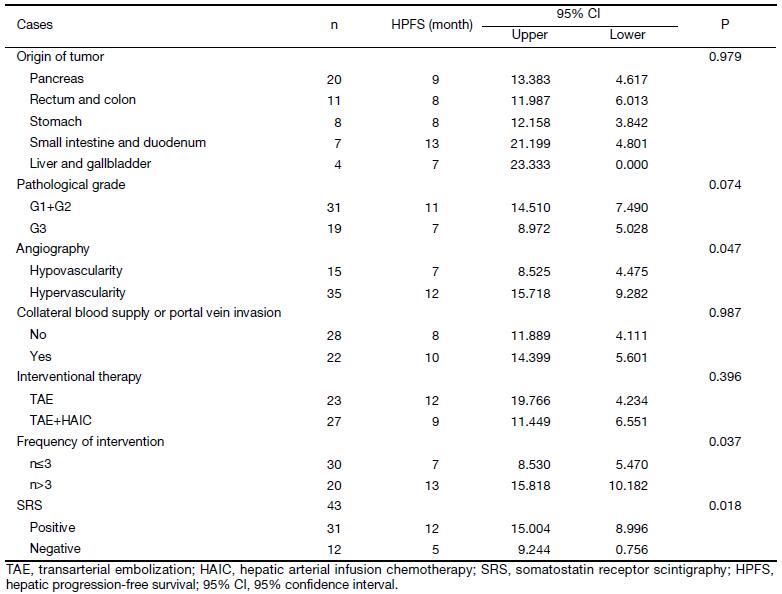
Among the origin of tumor, the pathological grade, angiography, vascular variation and frequency of interventional therapy, angiography (P=0.047) and frequency (P=0.037) showed significantly statistical differences (Table 1, Figure 2A, B). It means that the patients with rich angiography appearance or receiving more than 3 times of interventional treatment had longer HPFS.
Full table
Forty-three patients were examined with somatostatin receptor scintigraphy (SRS). The patients with positive results could get longer HPFS (P=0.018) (Table 1, Figure 2C).
In the Cox regression models, only frequency of intervention (n≤3/n>3) was an independent predictor of HPFS [χ2=4.095; P=0.043; HR=0.533, 95% confidence interval (95% CI): 0.287–0.989] (Figure 2B). The other variables were not statistically significant in these models. This analysis showed that more than 3 times of interventional therapy made sense for patients.
Side effects
The postoperative side effects included such tolerable reaction as nausea, vomiting, pain and myelosuppression. All side effects were 1–2 degrees, which could be relieved with symptomatic treatment. Two months after interventional treatment, there was one case of pulmonary embolism, two cases of diaphragmatic abscess and one case of pancreatitis. The above symptoms were alleviated after symptomatic treatment.
Discussion
For patients with GEP-NETs, it is critical to optimize palliative care for effective and efficient treatment of hepatic metastases. According to the current National Comprehensive Cancer Network (NCCN) and European Society of Medical Oncology (ESMO) consensus, interventional therapy is an important method for the palliative, multidisciplinary cooperative treatment of advanced hepatic metastasis from NETs based on systemic therapy. However, additional data are required concerning the methods, treatment protocols, chemotherapy and complications for interventional therapy.
Most of the hepatic metastases from NETs are found with abundant blood supply, approximately 90% of which are supplied by the hepatic artery. The theory provided a theoretical basis for the interventional treatment of blood vessels (12). We observed that the blood supply of most hepatic metastases was abundant (35/50) and primarily from the hepatic artery (13). Our analysis shows that patients with rich tumor staining had longer HPFS. We found 22 of the cases with collateral blood supply or portal vein invasion. Compared with 2.8%–8.1% of hepatic metastases from a common gastrointestinal tumor which had collateral blood supply, our findings were more serious in terms as progression (14,15). A poor treatment outcome will be caused by any lateral slip of the collateral blood supply, portal vein invasion, hepatic artery portal vein or hepatic venous fistula. The poor treatment outcome would lead to pulmonary embolism or other severe complications from an ectopic embolism. Admittedly, there were few studies on this topic. It may be related to the compensation of tumor blood vessels and tumor progression after repeated interventional therapies, and further exploration is required. The patients receiving more than 3 times of interventional treatment got longer HPFS, which means that multiple interventional therapies are required to achieve a satisfactory embolization effect.
At present, drug-loaded microspheres (Drug-Eluting, Beads, CalliSpheres) and Yttrium 90 radioactive microspheres were not comprehensively applied to check up blood vessels. Interventional treatment of blood vessels is mostly with vascular embolization. When the effect of multidisciplinary treatment was poor, or when the angiography showed a poorly stained tumor, HAIC should be added as a treatment (16). For general cases, no significant difference was found between TAE alone and TAE with HAIC. However, in cases of hypovascularity angiography, only those cases treated with HAIC had obtained the effect of PR.
We performed SRS examinations for some of the patients, and patients with the positive results were able to get a longer HPFS. Theoretically, the results of positive SRS correlated with hypervascular tumors (17).
For patients with hepatic metastases from NETs, it is commonly accepted that there is no difference between TAE and HAIC albeit clearly demonstrated (18,19). A consistent trend was observed in our cases even complications was widely found in HAIC (20,21). Patients who received HAIC suffered from pancreatitis and subphrenic abscess. As long as the discovery and treatment were timely, there would be no serious consequences.
As to patients received intervention as first- and second-line therapy, it was reported (12) that the valid control rates were 74% and 75%, respectively. When the intervention was performed as third-line and follow-up therapy, the valid control rates were 59% and 40%, respectively. In mostly relapsed cases of this study, interventional therapy was often used as a second- or third-line therapy. The overall response rate (64%) was similar to that of the third-line treatment. During the multidisciplinary treatment of GEP-NETs, especially in the cases with hepatic metastases as the main symptoms, we suggest that the blood supply of liver lesion should be examined by imaging, especially for those with positive SRS. The interventional therapy can be considered earlier. The number of treatment should be more than 3 times.
Conclusions
Vascular intervention therapy is safe and effective for patients with hepatic metastasis from GEP-NETs. In recent years, interventional therapy is widely selected to treat hypo- and hypervascularized hepatic tumors, and the therapeutic effect has been confirmed. It is sure that along with the gradual improvement of interventional therapy and achievements in clinical prospective trials, more treatment options will be offered based on interventional multidisciplinary collaborative therapy for NETs to achieve a more definitive treatment effect.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (No. 81571781).
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Oberg K. Neuroendocrine tumors (NETs): historical overview and epidemiology. Tumori 2010;96:797–801.
- Proye C. Natural history of liver metastasis of gastroenteropancreatic neuroendocrine tumors: place for chemoembolization. World J Surg 2001;25:685–8.
- Krausch M, Raffel A, Anlauf M, et al. " Cherry picking”, a multiple non-anatomic liver resection technique, as a promising option for diffuse liver metastases in patients with neuroendocrine tumours. World J Surg 2014;38:392–401. DOI:10.1007/s00268-013-2267-3
- Ito T, Sasano H, Tanaka M, et al. Epidemiological study of gastroenteropancreatic neuroendocrine tumors in Japan. J Gastroenterol 2010;45:234–43. DOI:10.1007/s00535-009-0194-8
- Jiao X, Li Y, Wang H, et al. Clinicopathological features and survival analysis of gastroentero-pancreatic neuroendocrine neoplasms: a retrospective study in a single center of China. Chin J Cancer Res 2015;27:258–66. DOI:10.3978/j.issn.1000-9604.2015.06.04
- Bruzoni M, Parikh P, Celis R, et al. Management of the primary tumor in patients with metastatic pancreatic neuroendocrine tumor: a contemporary single-institution review. Am J Surg 2009;197:376–81. DOI:10.1016/j.amjsurg.2008.11.005
- Alagusundaramoorthy SS, Gedaly R. Role of surgery and transplantation in the treatment of hepatic metastases from neuroendocrine tumor. World J Gastroenterol 2014;20:14348–58. DOI:10.3748/wjg.v20.i39.14348
- Burad DK, Kodiatte TA, Rajeeb SM, et al. Neuroendocrine neoplasms of liver — A 5-year retrospective clinic-pathological study applying World Health Organization 2010 classification. World J Gastroenterol 2016;22:8956–66. DOI:10.3748/wjg.v22.i40.8956
- Yao JC, Hassan M, Phan A, et al. One hundred years after " carcinoid”: epidemiology of and prognostic factors for neuroendocrine tumors in 35,825 cases in the United States. J Clin Oncol 2008;26:3063–72. DOI:10.1200/JCO.2007.15.4377
- Vogl TJ, Naguib NN, Zangos S, et al. Liver metastases of neuroendocrine carcinomas: interven-tional treatment via transarterial embolization, chemoembolization and thermal ablation. Eur J Radiol 2009;72:517–28. DOI:10.1016/j.ejrad.2008.08.008
- Kennedy AS. Hepatic-directed therapies in patients with neuroendocrine tumors. Hematol Oncol Clin North Am 2016;30:193–207. DOI:10.1016/j.hoc.2015.09.010
- de Baere T, Deschamps F, Tselikas L, et al. GEP-NETS update: Interventional radiology: role in the treatment of liver metastases from GEP-NETs. Eur J Endocrinol 2015;172:R151–66. DOI:10.1530/EJE-14-0630
- Ronot M, Cuccioli F, Dioguardi Burgio M, et al. Neuroendocrine liver metastases: Vascular patterns on triple-phase MDCT are indicative of primary tumour location. Eur J Radiol 2017;89:156–62. DOI:10.1016/j.ejrad.2017.02.007
- Zhang H, Guo J, Gao S, et al. Prognostic factors for transarterial chemoembolization combined with sustained oxaliplatin-based hepatic arterial infusion chemotherapy of colorectal cancer liver metastasis. Chin J Cancer Res 2017;29:36–44. DOI:10.21147/j.issn.1000-9604.2017.01.05
- Tada K, Kokudo N, Seki M, et al. Hepatic resection for colorectal metastasis with macroscopic tumor thrombus in the portal vein. World J Surg 2003;27:299–303. DOI:10.1007/s00268-002-6716-7
- Okubo Y, Motohashi O, Nakayama N, et al. The clinicopathological significance of angiogenesis in hindgut neuroendocrine tumors obtained via an endoscopic procedure. Diagn Pathol 2016;11:128. DOI:10.1186/s13000-016-0580-5
- Sergieva S, Robev B, Dimcheva M, et al. Clinical application of SPECT-CT with 99mTc-Tektrotyd in bronchial and thymic neuroendocrine tumors (NETs). Nucl Med Rev Cent East Eur 2016;19:81–7. DOI:10.5603/NMR.2016.0017
- Gupta S. Intra-arterial liver-directed therapies for neuroendocrine hepatic metastases. Semin Intervent Radiol 2013;30:28–38. DOI:10.1055/s-0033-1333651
- Fiore F, Del Prete M, Franco R, et al. Transarterial embolization (TAE) is equally effective and slightly safer than transarterial chemoembolization (TACE) to manage liver metastases in neuroendocrine tumors. Endocrine 2014;47:177–82. DOI:10.1007/s12020-013-0130-9
- Oberg K, Akerström G, Rindi G, et al. Neuroendocrine gastroenteropancreatic tumors: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol 2010;21Suppl 5:v223–7. DOI:10.1093/annonc/mdq192
- Kennedy A, Bester L, Salem R, et al. Role of hepatic intra-arterial therapies in metastatic neuroendocrine tumours (NET): guidelines from the NET-Liver-Metastases Consensus Conference. HPB (Oxford) 2015;17:29–37. DOI:10.1111/hpb.12326
