Coordinated transcription of ANRIL and P16 genes is silenced by P16 DNA methylation
Introduction
The P15-P14-P16 (INK4) gene locus is a significant genetic susceptibility locus in cancers and cardiovascular disease (1-3). It has also been linked to a number of other pathologies, including intracranial aneurysm, type-2 diabetes, periodontitis, Alzheimer’s disease, endometriosis, frailty in the elderly, and glaucoma (4-6). The results of the Cancer Genome Project showed that this locus is the most frequently deleted region in thousands of human cancer genomes (7). While P16 genetic deletions do occur in cancers, the frequency of P16 DNA methylation is three times that of deletions (8,9). P15, P14 and P16 genes coordinately control the G1→S transition in the cell cycle through the cyclin-dependent kinase (CDK) 2/4/6-cyclin D1/E-Rb pathway (10). Higher-order chromatin structure analysis showed that the P15-P14-P16 chromatin presents in a dynamic compact loop for coordinate repression of these genes through CTCF-binding and that the chromatin decompaction leads to activation of these genes (11,12).
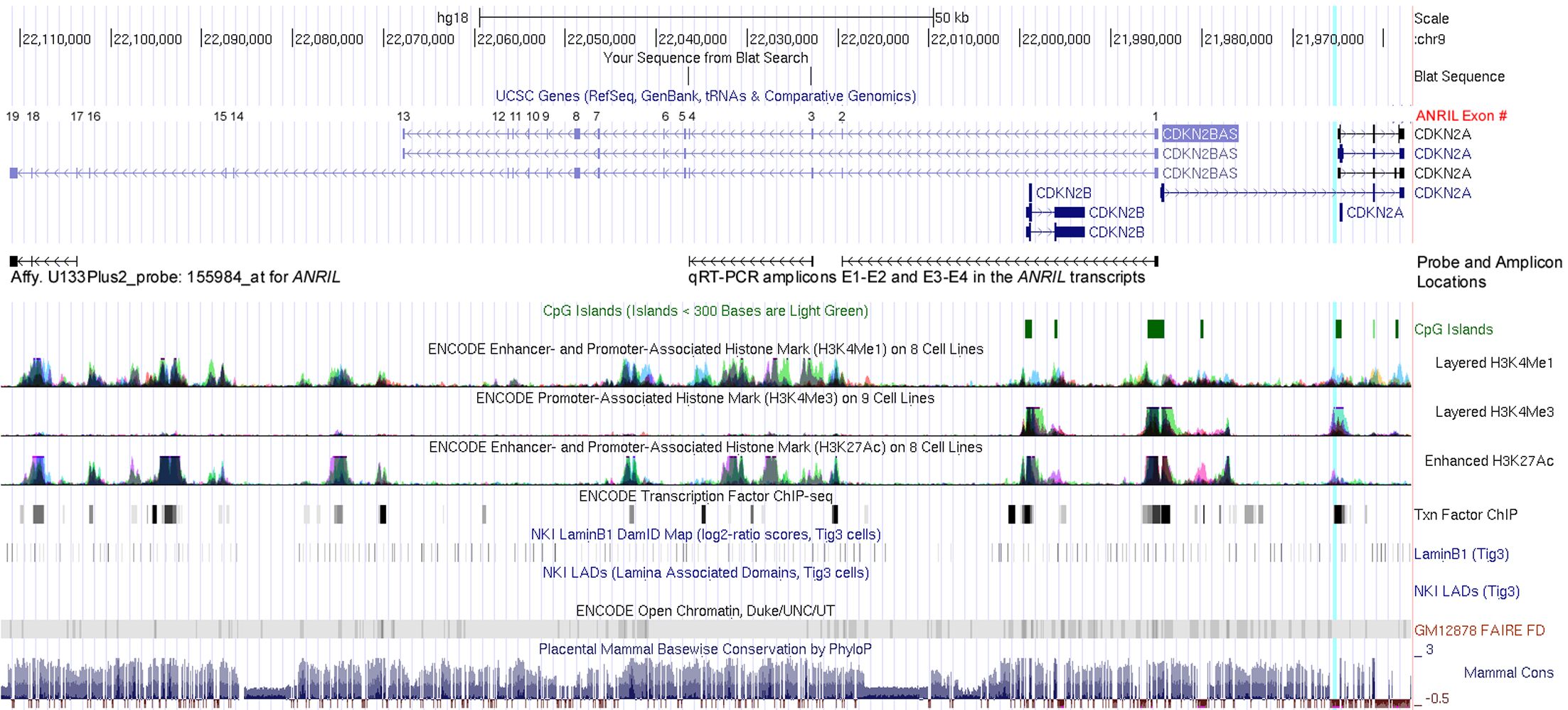
LncRNA ANRIL (CDKN2B-AS1) is transcribed from the antisense-strand of the P14 promoter and P15 gene regions in the INK4 locus (chr9: 21,994,791–22,120,544 in the hg19 human assembly) (13,14). The preliminary ANRIL transcripts are spliced into various mature forms (Supplementary Figure S1) (6,15). It was reported that ANRIL and miR31HG might downregulate P15 and P16 transcription through interacting with components of polycomb repressive complex-1 or -2 (PRC-1/-2) (16-19). However, the transcriptional relationship between these genes within the INK4 locus has not been well studied. The regulatory mechanism of ANRIL transcription has rarely been reported (20). In the present study, we report that P16 DNA methylation may repress ANRIL transcription. Bioinformatics and experimental analysis support that ANRIL and P16 are coordinately transcribed in human cells.
Materials and methods
Patients tissue collection
Colon carcinoma along with paired non-cancerous surgical margin samples were collected from 76 patients at the Peking University Cancer Hospital from 2009 to 2011. The Ethics Committee of Peking University Cancer Hospital approved this study. All patients provided written informed consent to participate in the study. The authors had no access to information that could identify individual participants during or after data collection.
Database sources
Gene expression and copy number of 995 cancer cell lines was downloaded from the publicly available database [https://portals.broadinstitute.org/ccle/home] of the Broad-Novartis Cancer Cell Line Encyclopedia (CCLE) (21).
Cell lines and culture
Human cell lines BGC823, MGC803, RKO, H1299, GES1 and SGC7901 were cultured in RPMI1640 medium supplemented with 10% fetal bovine serum (FBS). The human cell lines Caski, Colo205, HeLa, MHCC97H, Siha and SW480 were cultured in Dulbecco’s modified eagle medium (DMEM) with 10% FBS. AGS and PC3 were cultured in F12 medium with 10% FBS. All of these cell lines were cultured at 37 °C in 5% CO2. PC3 cell line was purchased from Cell Line Bank, Chinese Academy of Medical Science. RKO cell line was kindly provided by Dr. Guoren Deng at University of California San Francisco, and SW480 cell line, by Dr. Yuanjia Chen at Peking Union Medical College Hospital. AGS (from Dr. Chengchao Shou), GES1, BGC823, SGC7901, HeLa, Caski, Siha, H1299, Colo205 and MHCC97H cell lines (from Dr. Yang Ke) were obtained from laboratories at Peking University Cancer Hospital/Institute.
RNA extraction and reverse transcription-polymerase chain reaction (RT-PCR)
These cells were harvested when the density reached about 70%. Total RNA was extracted by TRIzol (Invitrogen) and reverse-transcribed using a first-cDNA synthesis kit (Transgen). The expression levels of the ANRIL, P16, P15 and P14 genes were analyzed by quantitative real-time polymerase chain reaction (qRT-PCR) using the corresponding primer sets (Table 1). To detect the linear ANRIL (NR_003529) expression status in cell lines and tissues, primer sets E3–E4 and E1–E2 were used for qRT-PCR and RT-PCR, respectively. Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) was used as a reference gene. Power SYBR Green PCR Master Mix (Fermentas) was used in qRT-PCR (ABI-7500). Relative mRNA level was calculated based on the average Ct value of the target gene and the GAPDH reference [

Full table
Transfection and construction of vectors
P16-specific siRNAs (5’-ccguaaauguccauuuauatt-3’ and 5’-uauaaauggacauuuacggtt-3’) were synthesized in GenePharma (Shanghai) and used to transiently transfect cells at a final concentration of 1.0 μg/mL. The negative control was scramble siRNAs (5’-uucuccgaacgugucacgutt-3’ and 5’-acgugacacguucggagaatt-3’) (GenePharma, Shanghai). The P16 expression vector was constructed by integrating the wild-type P16 cDNA into the pIRES2-EGFP vector. A total of 4.5×104 cells were transiently transfected with the pIRES2-P16 expression vector, seeded into each well (diameter, 3.48 cm; 6-well microplate; 3 wells/group), and incubated for 48 h. The P16-Dnmt_pTRIPZ and P16-ATF_pcDNA3.1 vectors were constructed as described previously (22,23). These experiments were repeated 2–3 times.
Detection of P16 and P14 methylation by denaturing high performance liquid chromatography (DHPLC)
The methylation status of the P16 and P14 CpG islands was determined using DHPLC as previously described (22,24,25).
Western blotting
Protein extracts were equally loaded into 12% acrylamide gels, electrophoresed, and transferred to PVDF membranes (Millipore). After blocking with 5% fat-free milk in PBS, the membranes were probed with rabbit anti-P16 (Ab50282; Abcam) and mouse anti-GAPDH (60004-1-Ig; Proteintech) which were diluted at 1:3,000 and 1:10,000, respectively. After incubation at room temperature for 1 h, membranes were washed with PBST three times, and incubated with horse radish peroxidase (HRP)-labeled secondary antibody goat anti-rabbit IgG (1:5,000) and anti-mouse IgG (1:10,000), respectively. Signals were visualized using the Enhanced Chemiluminescence kit (Pierce).
Statistical analysis
All statistical analyses were performed using SPSS 18.0 software (SPSS Inc., Chicago, IL, USA). Student’s t-test was conducted for normally distributed data. The significance of gene expression associations (r) was statistically tested with adjusting gene copy number, and P<0.05 was considered statistically significant.
Results
Transcription of ANRIL is significantly correlated with P16 expression
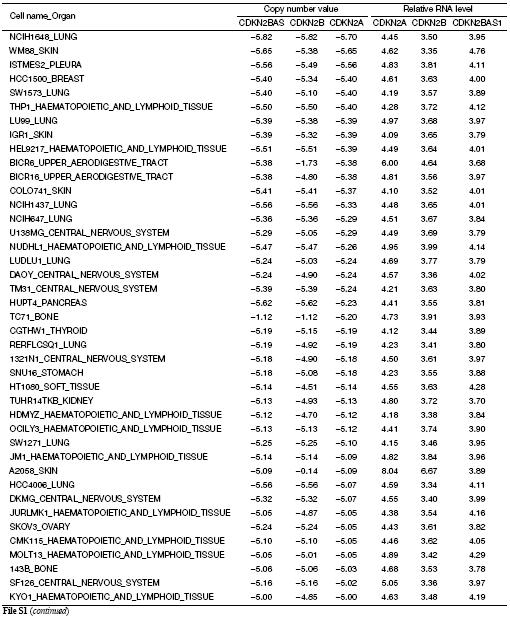
In order to investigate the relationship between transcription levels of the genes within the INK4 locus, we analyzed the publicly available databases in the CCLE project (21). The INK4 locus is absent from 298 cell lines [copy number value (CN) <–2.0] and is present in 713 cell lines ( Supplementary File S1). Because the 298 INK4 locus-absent cell lines cannot express the locus genes (Figure 1A), only the INK4 allele-containing cell lines were used in the correlation analysis. Unexpectedly, we found that the transcription level of ANRIL was positively and significantly correlated with the transcription level of P16 in the 713 cell lines (rs=0.254, P<0.001), but was not correlated with transcription levels ofP15 (rs=–0.015, P=0.701), although the P16 and P15 expression levels were correlated with each other (Figure 1B). P16 mRNA levels were still positively correlated with the ANRIL expression levels after the gene copy number was adjusted (P<0.001). Among 224 cell lines without theINK4 locus deletion (CN>0.0), the same phenomenon was consistently observed (Figure 1C). These results indicated that ANRIL is coordinately transcribed with P16 but not with P15.
Full table

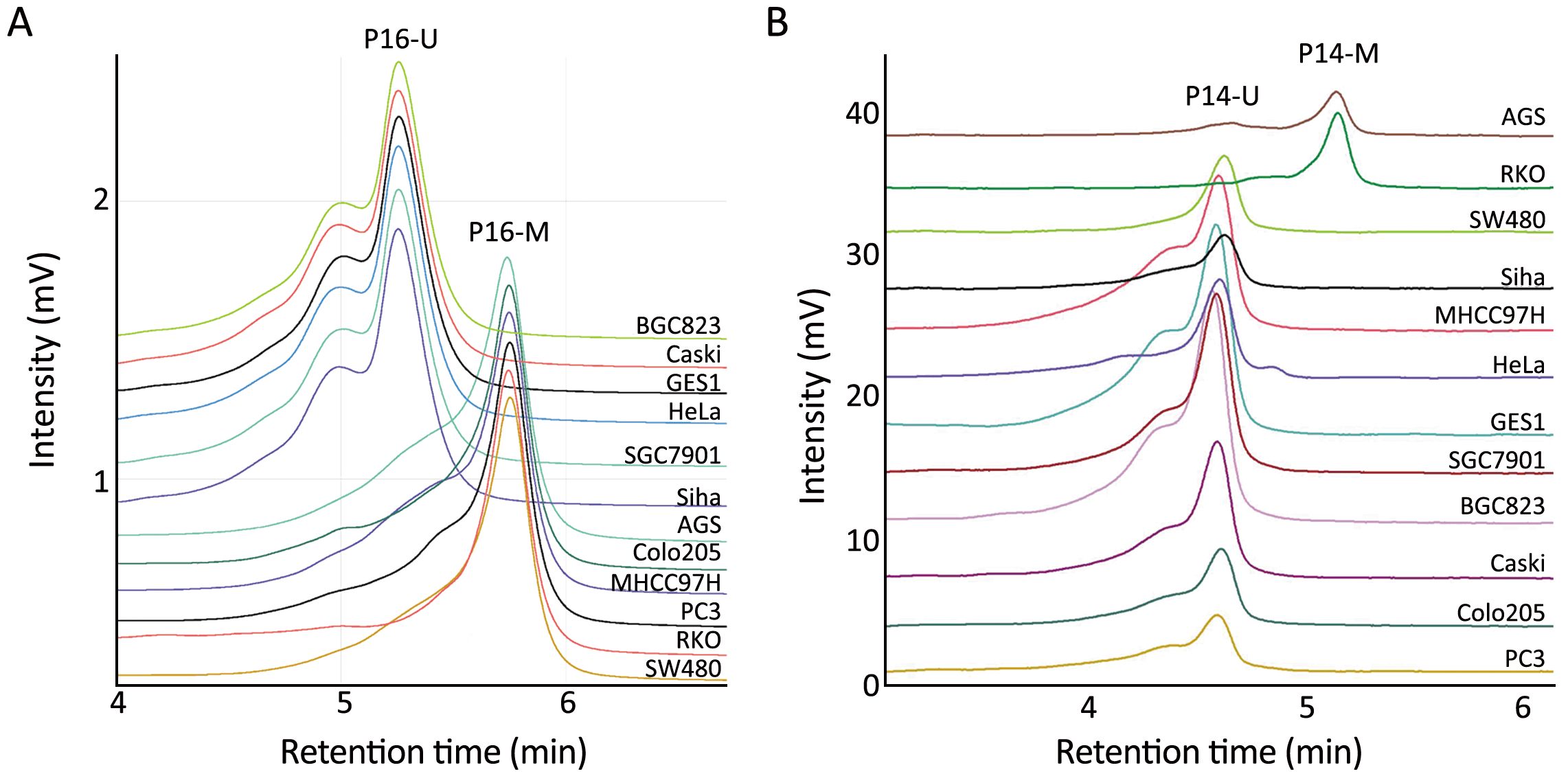
To confirm these correlations, we analyzed the transcription levels of ANRIL, P16, P15 and P14 genes in a panel of cell lines without the INK4 deletion using qRT-PCR, and observed the same phenomena. As expected, high level of ANRIL transcription was only observed in the P16-unmethylated cell lines (Caski, SGC7901, BGC823, GES1, Siha and HeLa), but not in P16-methylated cell lines (Colo205, PC3, MHCC97H, RKO, SW480 and AGS) (Figure 2A, 3A, 3B). P16 mRNA expression level was closely correlated with ANRIL expression level (r=0.774, P=0.003), suggesting that P16 DNA methylation might repress transcription of ANRIL. Such an association was not observed between the ANRIL level and the P15 or P14 mRNA levels (r=0.090 or 0.297, P=0.780 or 0.349) (Figure 3C, D).

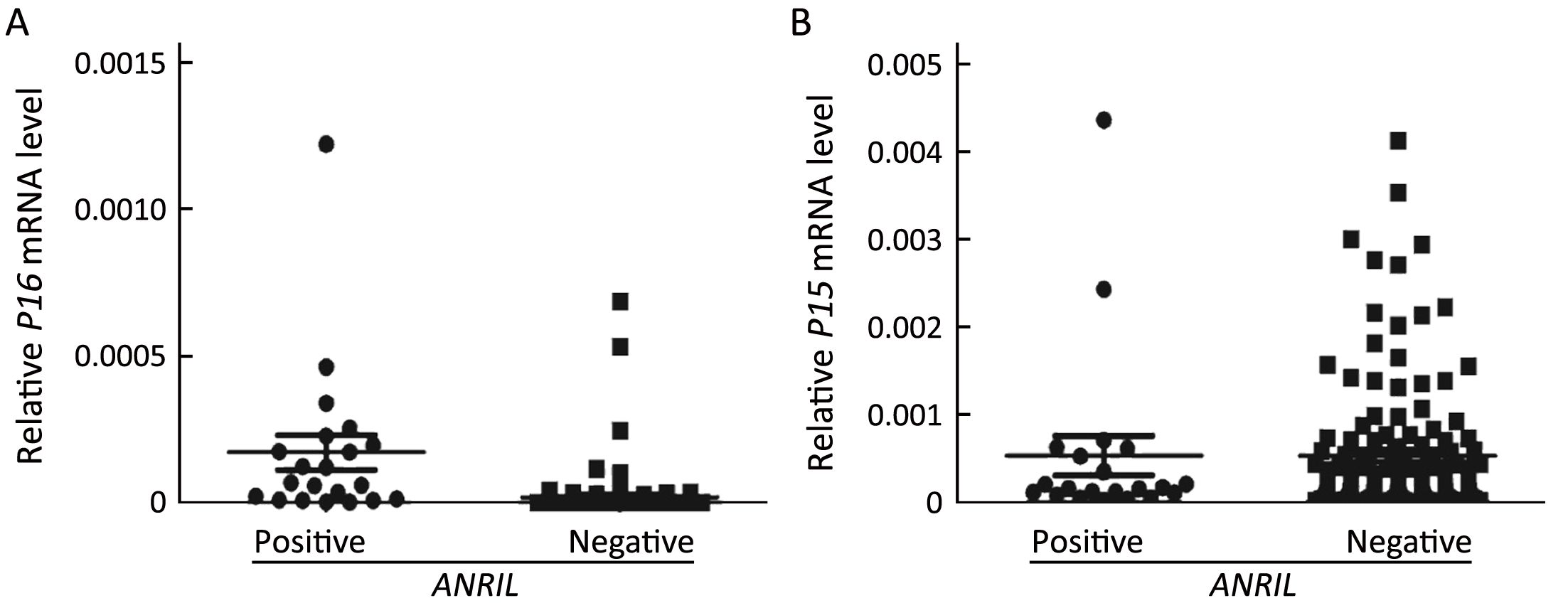
The positive relationship between the ANRIL and P16 expression levels was further validated in human colon tissue samples. As expected, the ANRIL expression level was significantly increased in colon cancer tissues compared to corresponding non-cancerous surgical margins [χ2 test, 19/76 (25.0%) vs. 2/76 (2.6%), P<0.001]. No significant association was observed between theANRIL lncRNA expression level and pTNM stage or other clinico-pathological characteristics or overall survival of colon cancer patients (data not shown). Notably, the P16 mRNA level was significantly higher in the ANRIL-positive samples than in the ANRIL-negative samples (P<0.001;Figure 4A). Once again, the P15 mRNA expression level was similar to the ANRIL-positive samples compared to the ANRIL-negative samples (P=0.879; Figure 4B). These results indicated that ANRIL and P16 genes are coordinately transcribed in human cells.
Interestingly, the CpG islands around the P14 transcription start site were methylated only in the AGS and RKO cells but not in the remaining cells (Figure 2B). Because the number of the P14-methylated cell lines is limited, an association between ANRIL transcription and P14 methylation was not observed.
Transcription repression of ANRIL by P16 DNA methylation
Recently, we constructed a P16 promoter DNA methyltransferase (P16-Dnmt) that can specifically methylate P16 CpG islands around transcription start sites, and this subsequently leads to silencing of P16 transcription in human cells but cannot induce P14 methylation or P14 repression (20). To elucidate whether methylation of P16 CpG islands directly leads to the silencing of ANRIL transcription, we analyzed the transcription level of ANRIL in GES1 and BGC823 cells, in which P16 CpG islands were methylated by the stably transfected P16-Dnmt_pTRIPZ vector with induction of 0.25 μg/mL doxycycline for 14 days (Figure 5A, C). qRT-PCR analysis showed that the transcriptional activities of both P16 and ANRIL were significantly decreased, whereas the P15 mRNA expression level was not changed, and the P14 mRNA expression level was slightly increased (Figure 5B, D) in these cell lines.

By contrast, using a P16-specific artificial transcription factor (P16-ATF) that can induce demethylation of P16 CpG islands and reactivate gene expression (Figure 5E) (23), we found that transcriptional activities of both P16 and ANRIL were significantly increased (P<0.001), whereasP15 and P14 mRNA expression levels were not changed in H1299 cells that were stably transfected with the P16-ATF_pcDNA3.1 vector (Figure 5F). These results demonstrated that P16 DNA methylation indeed represses ANRIL transcription.
Transcription of ANRIL is not affected by changes in P16 expression
To clarify whether downregulation of ANRIL expression is directly caused by P16 DNA methylation itself or indirectly caused by subsequent P16 repression, we induced P16 expression changes in H1299 and BGC823 cells through transient transfection, and found that the ANRIL level remained stable following P16 overexpression and siRNA knockdown (Figures 6A, B), respectively. Similar result was also obtained in MGC803 cells (Figure 6C). These data suggest that it is P16 methylation itself, but not P16 downregulation, that leads to the repression of ANRIL transcription.

Discussion
ANRIL is transcribed from the INK4 locus and can be spliced into diverse mature forms to regulate gene transcription in the nucleus and to interact with proteins in the cytoplasm (6,15). Consistent with others’ reports that ANRIL is upregulated in many cancers (26-32), we find that ANRIL is also significantly upregulated in colon cancer. The mechanisms of regulation of ANRIL transcription have rarely been reported (20). In the present study, we report that the endogenous ANRIL and P16 genes are coordinately transcribed in cancer cells in vitro and in vivo, and that P16 methylation may repress ANRIL transcription.
ANRIL is considered to be an oncogene, according to its frequent upregulation in many cancers and association with a poor prognosis (26-32). In the present study, we observed significant ANRIL upregulation in colon cancer tissues. There was no association between the ANRIL lncRNA expression level and prognosis of colon cancer.
There are a number of regulators that co-regulate transcription of P15, P14 and P16 (10,17,33). It is reasonable to expect that ANRIL lncRNA expression correlates with that of the genes in the INK4 locus. In contrast to another report that ANRIL (CDKN2B-AS) may repress P15 expression (34), we did not observe any correlation between the expression levels of ANRIL and P15, both in a large number of cancer cell lines and in colon tissues. Interestingly, we found a strong positive relationship between the expression levels of ANRIL and P16. Our experimental evidence strongly supports that the methylation status of the P16 CpG islands directly affects the transcriptional activity of both ANRIL and P16, indicating the bi-directional functions of the P16 promoter. It was previously reported that P16 methylation was associated with loss of CTCF binding and a chromatin boundary in the P16 gene (11). The INK4 chromatin is enriched with the CTCF binding sites and CTCF depletion disrupts the INK4 chromatin loop formation (12). P16 methylation may repress ANRIL transcription via the INK4 chromatin conformation changes.
It was reported that ANRIL downregulation by siRNA increased the P16 expression level in cancer cells (18). However, an inverse relationship between the ANRIL and P16 transcription levels was not observed in 224 cancer cell lines without P16 allele deletion and colon cancer tissues from 76 patients. Instead, higher P16 mRNA expression was detected in cell lines and tissues with ANRIL expression than those without ANRIL expression. Endogenous ANRIL and P16 expression levels are positively correlated with each other in vitro and in vivo.
P16 is a weakly expressed nucleic protein that inhibits CDK4 activity in normal human cells (1). Although P16 expression is epigenetically repressed in approximately 30%–40% of cancers (8,9,35-38), strong cytoplasmic P16 staining could be detected in some cancers, especially in the human papillomavirus (HPV) infection-related cancers (i.e., cervical, oral, skin and thyroid cancers) (39-42). Interestingly, according to the publicly available database in the Human Protein Atlas project, the majority of P16 proteins are incorrectly located in the cytoplasm of P16 staining-positive cancer cells from multiple cancer types (i.e., breast, colon, gliocyte, liver, lung, melanocyte, ovarian, prostate, stomach, and urothelial tract), where HPV infection has not been reported (Supplementary Figure S2 ) (43). Cytoplasmic P16 cannot function as a tumor suppressor in cells. It is worth studying whether the cytoplasmic compartmentalization of P16 in cancer cells leads to adaptive upregulation of ANRIL transcription in cancer cells.

Conclusions
ANRIL is coordinately transcribed with P16 in human cells. The methylation status of the P16 CpG islands around the transcription start site controls the transcription of the ANRIL and P16 genes.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (No. 91640108); and the 973 Program of China (No. 2015CB553902). We thank Jacqueline Zayas (Mayo Clinic Graduate School of Biomedical Sciences, Rochester, MN 55905, USA) for English language editing.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Serrano M, Hannon GJ, Beach D. A new regulatory motif in cell-cycle control causing specific inhibition of cyclin D/CDK4. Nature 1993;366:704–7. [PubMed] DOI:10.1038/366704a0
- Kamb A, Gruis NA, Weaver-Feldhaus J, et al. A cell cycle regulator potentially involved in genesis of many tumor types. Science 1994;264:436–40. [PubMed]
- Kamijo T, Zindy F, Roussel MF, et al. Tumor suppression at the mouse INK4a locus mediated by the alternative reading frame product p19ARF. Cell 1997;91:649–59. [PubMed] DOI:10.1016/S0092-8674(00)80452-3
- Freedberg DE, Rigas SH, Russak J, et al. Frequent p16-independent inactivation of p14ARF in human melanoma. J Natl Cancer Inst 2008;100:784–95. [PubMed] DOI:10.1093/jnci/djn157
- Harismendy O, Notani D, Song X, et al. 9p21 DNA variants associated with coronary artery disease impair interferon-γ signalling response. Nature 2011;470:264–8. [PubMed] DOI:10.1038/nature09753
- Holdt LM, Stahringer A, Sass K, et al. Circular non-coding RNA ANRIL modulates ribosomal RNA maturation and atherosclerosis in humans. Nat Commun 2016;7:12429. [PubMed] DOI:10.1038/ncomms12429
- Beroukhim R, Mermel CH, Porter D, et al. The landscape of somatic copy-number alteration across human cancers. Nature 2010;463:899–905. [PubMed] DOI:10.1038/nature08822
- Merlo A, Herman JG, Mao L, et al. 5’ CpG island methylation is associated with transcriptional silencing of the tumour suppressor p16/CDKN2/MTS1 in human cancers. Nat Med 1995;1:686–92. [PubMed]
- Deng D, Liu Z, Du Y. Epigenetic alterations as cancer diagnostic, prognostic, and predictive biomarkers. Adv Genet 2010;71:125–76. [PubMed] DOI:10.1016/B978-0-12-380864-6.00005-5
- Gil J, Peters G. Regulation of the INK4b-ARF-INK4a tumour suppressor locus: all for one or one for all. Nat Rev Mol Cell Biol 2006;7:667–77. [PubMed] DOI:10.1038/nrm1987
- Witcher M, Emerson BM. Epigenetic silencing of the p16INK4a tumor suppressor is associated with loss of CTCF binding and a chromatin boundary. Mol Cell 2009;34:271–84. [PubMed] DOI:10.1016/j.molcel.2009.04.001
- Hirosue A, Ishihara K, Tokunaga K, et al. Quantitative assessment of higher-order chromatin structure of the INK4/ARF locus in human senescent cells. Aging Cell 2012;11:553–6. [PubMed] DOI:10.1111/j.1474-9726.2012.00809.x
- Pasmant E, Laurendeau I, Héron D, et al. Characterization of a germ-line deletion, including the entire INK4/ARF locus, in a melanoma-neural system tumor family: identification of ANRIL, an antisense noncoding RNA whose expression coclusters with ARF. Cancer Res 2007;67:3963–9. [PubMed] DOI:10.1158/0008-5472.CAN-06-2004
- Yu W, Gius D, Onyango P, et al. Epigenetic silencing of tumour suppressor gene p15 by its antisense RNA. Nature 2008;451:202–6. [PubMed] DOI:10.1038/nature06468
- Burd CE, Jeck WR, Liu Y, et al. Expression of linear and novel circular forms of an INK4/ARF-associated non-coding RNA correlates with atherosclerosis risk. PLoS Genet 2010;6:e1001233. [PubMed] DOI:10.1371/journal.pgen.1001233
- Aguilo F, Zhou MM, Walsh MJ. Long noncoding RNA, polycomb, and the ghosts haunting INK4b-ARF-INK4a expression. Cancer Res 2011;71:5365–9. [PubMed] DOI:10.1158/0008-5472.CAN-10-4379
- Yap KL, Li S, Muñoz-Cabello AM, et al. Molecular interplay of the Non-coding RNA ANRIL and methylated histone H3 lysine 27 by Polycomb CBX7 in transcriptional silencing of INK4a. Mol Cell 2010;38:662–74. [PubMed] DOI:10.1016/j.molcel.2010.03.021
- Xu S, Wang H, Pan H, et al. ANRIL lncRNA triggers efficient therapeutic efficacy by reprogramming the aberrant INK4-hub in melanoma. Cancer Lett 2016;381:41–8. [PubMed] DOI:10.1016/j.canlet.2016.07.024
- Montes M, Nielsen MM, Maglieri G, et al. The lncRNA MIR31HG regulates p16INK4A expression to modulate senescence. Nat Commun 2015;6:6967. [PubMed] DOI:10.1038/ncomms7967
- Wan G, Mathur R, Hu X, et al. Long non-coding RNA ANRIL (CDKN2B-AS) is induced by the ATM-E2F1 signaling pathway. Cell Signal 2013;25:1086–95. [PubMed] DOI:10.1016/j.cellsig.2013.02.006
- Cancer Cell Line Encyclopedia Consortium; Genomics of Drug Sensitivity in Cancer Consortium. Pharmacogenomic agreement between two cancer cell line data sets. Nature 2015;528:84–7. [PubMed] DOI:10.1038/nature15736
- Cui C, Gan Y, Gu L, et al. P16-specific DNA methylation by engineered zinc finger methyltransferase inactivates gene transcription and promotes cancer metastasis. Genome Biol 2015;16:252. [PubMed] DOI:10.1186/s13059-015-0819-6
- Zhang B, Xiang S, Zhong Q, et al. The p16-specific reactivation and inhibition of cell migration through demethylation of CpG islands by engineered transcription factors. Human Gene Ther 2012;23:1071–81. [PubMed] DOI:10.1089/hum.2012.070
- Deng D, Deng G, Smith MF, et al. Simultaneous detection of CpG methylation and single nucleotide polymorphism by denaturing high performance liquid chromatography. Nucleic Acids Res 2002;30:E13. [PubMed]
- Luo D, Zhang B, Lv L, et al. Methylation of CpG islands of p16 associated with progression of primary gastric carcinomas. Lab Invest 2006;86:591–8. [PubMed] DOI:10.1038/labinvest.3700415
- Zhang D, Sun G, Zhang H, et al. Long non-coding RNA ANRIL indicates a poor prognosis of cervical cancer and promotes carcinogenesis via PI3K/Akt pathways. Biomed Pharmacother 2017;85:511–6. [PubMed] DOI:10.1016/j.biopha.2016.11.058
- Zou ZW, Ma C, Medoro L, et al. LncRNA ANRIL is up-regulated in nasopharyngeal carcinoma and promotes the cancer progression via increasing proliferation, reprograming cell glucose metabolism and inducing side-population stem-like cancer cells. Oncotarget 2016;7:61741–54. [PubMed] DOI:10.18632/oncotarget.11437
- Sun Y, Zheng ZP, Li H, et al. ANRIL is associated with the survival rate of patients with colorectal cancer, and affects cell migration and invasion in vitro. Mol Med Rep 2016;14:1714–20. [PubMed] DOI:10.3892/mmr.2016.5409
- Qiu JJ, Wang Y, Liu YL, et al. The long non-coding RNA ANRIL promotes proliferation and cell cycle progression and inhibits apoptosis and senescence in epithelial ovarian cancer. Oncotarget 2016;7:32478–92. [PubMed] DOI:10.18632/oncotarget.8744
- Zhu H, Li X, Song Y, et al. Long non-coding RNA ANRIL is up-regulated in bladder cancer and regulates bladder cancer cell proliferation and apoptosis through the intrinsic pathway. Biochem Biophys Res Commun 2015;467:223–8. [PubMed] DOI:10.1016/j.bbrc.2015.10.002
- Lin L, Gu ZT, Chen WH, et al. Increased expression of the long non-coding RNA ANRIL promotes lung cancer cell metastasis and correlates with poor prognosis. Diagn Pathol 2015;10:14. [PubMed] DOI:10.1186/s13000-015-0247-7
- Zhang EB, Kong R, Yin DD, et al. Long noncoding RNA ANRIL indicates a poor prognosis of gastric cancer and promotes tumor growth by epigenetically silencing of miR-99a/miR-449a. Oncotarget 2014;5:2276–92. [PubMed] DOI:10.18632/oncotarget.1902
- Deng D, Li Q, Wang X. Methylation and demethylation of Ink4 locus in cancer development. Chin J Cancer Res 2010;22:245–52. DOI:10.1007/s11670-010-0245-y
- Kotake Y, Nakagawa T, Kitagawa K, et al. Long non-coding RNA ANRIL is required for the PRC2 recruitment to and silencing of p15INK4B tumor suppressor gene. Oncogene 2011;30:1956–62. [PubMed] DOI:10.1038/onc.2010.568
- Li Q, Wang X, Lu Z, et al. Polycomb CBX7 directly controls trimethylation of histone H3 at lysine 9 at the p16 locus. PLoS One 2010;5:e13732. [PubMed] DOI:10.1371/journal.pone.0013732
- Sun Y, Deng D, You WC, et al. Methylation of p16 CpG islands associated with malignant transformation of gastric dysplasia in a population-based study. Clin Cancer Res 2004;10:5087–93. [PubMed] DOI:10.1158/1078-0432.CCR-03-0622
- Cao J, Zhou J, Gao Y, et al. Methylation of p16 CpG island associated with malignant progression of oral epithelial dysplasia: a prospective cohort study. Clin Cancer Res 2009;15:5178–83. [PubMed] DOI:10.1158/1078-0432.CCR-09-0580
- Liu H, Liu XW, Dong G, et al. P16 methylation as an early predictor for cancer development from oral epithelial dysplasia: a double-blind multicentre prospective study. EBioMedicine 2015;2:432–7. [PubMed] DOI:10.1016/j.ebiom.2015.03.015
- Mooren JJ, Gültekin SE, Straetmans JM, et al. P16INK4A immunostaining is a strong indicator for high-risk-HPV-associated oropharyngeal carcinomas and dysplasias, but is unreliable to predict low-risk-HPV-infection in head and neck papillomas and laryngeal dysplasias. Int J Cancer 2014;134:2108–17. [PubMed] DOI:10.1002/ijc.28534
- Hoffmann M, Ihloff AS, Görögh T, et al. p16INK4a overexpression predicts translational active human papillomavirus infection in tonsillar cancer. Int J Cancer 2010;127:1595–602. [PubMed] DOI:10.1002/ijc.25174
- Carozzi F, Gillio-Tos A, Confortini M, et al. Risk of high-grade cervical intraepithelial neoplasia during follow-up in HPV-positive women according to baseline p16-INK4A results: a prospective analysis of a nested substudy of the NTCC randomised controlled trial. Lancet Oncol 2013;14:168–76. [PubMed] DOI:10.1016/S1470-2045(12)70529-6
- Steinestel J, Al Ghazal A, Arndt A, et al. The role of histologic subtype, p16INK4a expression, and presence of human papillomavirus DNA in penile squamous cell carcinoma. BMC Cancer 2015;15:220. [PubMed] DOI:10.1186/s12885-015-1268-z
- Uhlén M, Fagerberg L, Hallström BM, et al. Tissue-based map of the human proteome. Science 2015;347:1260419. [PubMed] DOI:10.1126/science.1260419
